Neobvyklý případ krvácení ze zažívacího traktu
Igor Tozzi di Angelo Orcid.org 1, Tomáš Malý Orcid.org 2, Čestmír Neoral Orcid.org 2, Jiří Kysučan Orcid.org , Bronislav Klementa Orcid.org 3, Martin Köcher Orcid.org 4, Michal Konečný Orcid.org 5
+ Pracoviště
Souhrn
Penetrující aortální aterosklerotický vřed je mezi akutními aortálními syndromy považován za fatální. Popisujeme případ nemocného s bolestmi v epigastriu, který zemřel v důsledku penetrujícího aortálního vředu hrudní aorty, který byl komplikován intramurálním hematomem středního jícnu, jenž se provalil do jícnu jako aortoezofageální píštěl a vedl k vykrvácení. V literatuře jsme našli pouze dva podobné případy.
Klíčová slova: aortální syndrom - aterosklerotický vřed - hematom - jícen.
THE CASE
58 years old male was admitted to the hospital because of a 3 days abdominal pain in the epigastric region, without propagation, nausea and vomiting He did not notice black stools or blood in stools. He was treated for a duodenal ulcer one year ago. He had chronic renal failure and was undergoing dialysis program due to polycystic kidneys. He was also treated for arterial hypertension and ischemic disease of the lower limbs.
His medication was Apo allupurinol, Betaloc, Furosemid, Rocaltrol and Vit.D.
In physical examination the only finding was palpable epigastric pain without any other pathological signs or findings. There were no signs of edema or ascites. Rectal examination revealed no bleeding. Laboratory findings: sedimentation rate 81 per hour, leukocytes were elevated 14.10-3 Red blood cell count was within normal. ranges, there was a slightly decreasing level of hemoglobin 125 g/l - 100 g/l. Platelets and coagulation parameters were within normal rates. Urea 12.4 mmol/l, creatinine 593 µmol/l. Liver function tests were normal.
Upper endoscopy was performed on the day of admission with the following findings: Submucosal lesion in the middle esophagus prominent and protruding to 1/3 of the lumen, deformation of the first part of duodenum without signs of ulceration, inflammation or acute bleeding. Endosongraphy and CT were recommended.
Figure l was similar to our endoscopic finding but this picture is not for the patient. This figure is copied from Gastrosource web site. (Submucosal lesions of esophagus).
On the next day of admission the patient had severe abdominal pain in the epigastric region without decrease in Hb or Er. Oriented, conscious with tachycardia 130/min. In a few hours the patient suddenly lost consciousness, pulse was not palpable, pale, transferred immediately to the intensive care unit where CPR was started and successfully the patient returns to be conscious, BP 120/80, O2 saturation 100% and complains of epigastric abdominal pain. After one hour the patient lost consciousness, ECG showed asystole, Nasogastric tube was full of fresh blood. CPR started again which was unsuccessful this time. The patient died.
The autopsy revealed perforation of the descending aorta with the severe ulcerative sclerotic plate into the mediastinum and the esophageal wall and exsanguination.
DISCUSSION
The four major acute aortic syndromes include aortic rupture, aortic dissection, intramural hematoma and penetrating atherosclerotic ulcer. Aortic dissection is caused by a circumferential or, less frequently, transverse tear of the intima. It often occurs along the right lateral wall of the ascending aorta where the hydraulic shear stress is high. Another common site is the descending thoracic aorta just below the ligamentum arteriosum. The initiating event is either a primary intimal tear with secondary dissection into the media or a medial hemorrhage that dissects into and disrupts the intima. The pulsatile aortic flow then dissects along the elastic lamellar plates of the aorta and creates a false lumen. The dissection usually propagates distally down the descending aorta and into its major branches, but it may also propagate proximally. Distal propagation may be limited by atherosclerotic plaque. In some cases, a secondary distal intimal disruption occurs, resulting in the reentry of blood from the false to the true lumen.
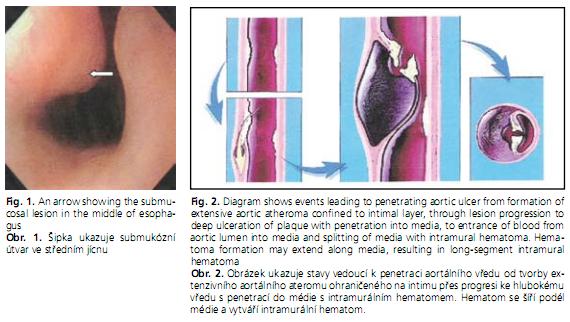
In a penetrating aortic ulcer, an atheromatous plaque ulcerates and disrupts the internal elastic lamina, burrowing deeply through the intima into the aortic media(1,2). When an atherosclerotic plaque penetrates into the media, the media is exposed to pulsatile arterial flow, which causes hemorrhage into the wall that then leads to intramural hematoma(3) (Figure 2). The plaque may precipitate a localized intramedial dissection associated with a variable amount of hematoma within the aortic wall, may break through into the adventitia to form a pseudoaneurysm, or may rupture. Ulceration of an aortic atheroma occurs in patients with advanced atherosclerosis. On imaging, a penetrating aortic ulcer can be distinguished from an atheromatous plaque by presence of a focal, contrast-filled outpouching surrounded by an intramural hematoma which confirms the aggressive behavior of the lesion. The atheromatous plaque with ulceration but without penetration through the intima shows irregular margins, but no contrast material extends beyond the level of intima, which is frequently calcified, and no intramural hematoma is present.
Eighty-seven percent of aortoesophageal fistulas (AEFs)(4) were the result of a thoracic aortic aneurysm, foreign body ingestion, or esophageal malignancy. The remaining cases of AEF were caused by surgical complication, esophageal reflux, traumatic false aneurysm, tuberculosis, corrosive esophagitis, congenital anomaly, instrumentation, or atherosclerotic disease. Only two cases of a penetrating atherosclerotic aortic ulcer rupturing into the thoracic esophagus have been reported(5) and ours is the third such report. The triad of mid thoracic pain, sentinel arterial hemorrhage, and final exsanguination after a symptom free interval has been termed as Chiari‘s triad(6). The latent interval between prodromal hemorrhage and exsanguination is unpredictable and ranges from 2 hours to 18 days (mean, 2 to 3 days)(7). A differential diagnosis considering the possibility of a thoracic aortic aneurysm should always be required in patients with dysphagia. Emergency surgery should always be considered in patients showing three characteristic symptoms (hematemesis, dysphagia, chest pain) before the occurrence of massive hemorrhage(8). A definitive diagnosis can be established when ulceration accompanied by coagulations are detected by endoscopy. Contrast enhanced CT scan can confirm the diagnosis(8). On account of high probability of death from exsanguination or from infection of the surrounding tissues and subsequent sepsis, an aggressive treatment without delay has been advised(8).
There is a little consensus about the optimal management of the condition. Several types of treatment have been described including open surgery, temporary control measures such as percutaneous embolization, and the use of a Sengstaken-Blakemore tube and more recently endovascular treatment(8).
Abbreviations
ACT - Activated Clotting Time
AEF - Aortoesophageal fistula
CP - Cardio Pulmonary
CPR - Cardiopulmonary Resucitation
PVC - Poly Vinyl Chloride
REFERENCES
- 1. Vilacosta I, San Roman JA. Acute aortic syndrome. Heart 2001; 85: 365-368.
- 2. Hayashi H, Matsuoka Y, Sakamoto I, et al. Penetrating atherosclerotic ulcer of the aorta: imaging features and disease concept. RadioGraphics 2000; 20: 995-1005.
- 3. Yasuda F, Shimono T, Tonouchi H, Shimpo H, Yada. Successful repair of an Aortoesophageal Fistula with Aneurysm From Esophageal Diverticulum. Ann Thorac Surg 2002; 73: 637-639.
- 4. Tolis G. Jr, Ramachandra CR, Valavanur A Subramanian. Repair of aortoesophageal fistula arising from a penetrating atherosclerotic ulcer of the distal transverse arch, CTSNET, 16 March.
- 5. Chiari H. Ueber Fremdkoreverletzung des Oesophagus mit Aortenperforation. Ber Klin Wochenschr 1914; 51: 7-9.
- 6. Yamada T, Sato H, Seki M, Kitagawa S, Nakagawa M, Shimazaki H. Succesful salvage lvage of Aortoesophageal Fistula Caused by a Fish Bone. Ann Thorac Surg 1996; 61: 1843-1845.
- 7. Unosawa S, Akiyama K, Nakata K, Masuda H, Wakabayashi K, Shiratori R, Kaneko Y, Negishi N, Sezai Y. Successful Surgical Treatment of an Aortoesophageal Fistula due to a Descending Aortic Aneurysm. Ann Thorac Cardiovasc Surg 2003; 9: 241-242.
- 8. Flores J, Shiiya N, Kunihara T, Yoshimoto K, Yasuda K. Aortoesophageal fistula: Alternatives of treatment; Case report and literature review. Ann Thorac Cardiovasc Surg 2004; 10: 241-246.
Pro přístup k článku se, prosím, registrujte.
Výhody pro předplatitele
Výhody pro přihlášené