Abstrakt
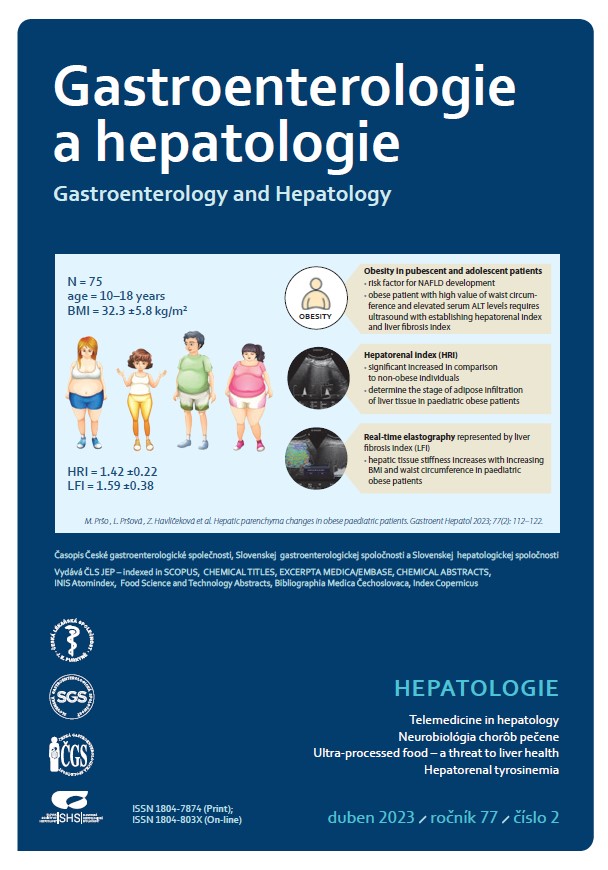
Úvod: Nealkoholová steatóza pečene (non-alcoholic fatty liver disease – NAFLD) predstavuje narastajúci klinický problém už v detstve a v adolescencii. Súčasné poznatky o NAFLD naznačujú dôležitú úlohu genetických a environmentálnych rizikových faktorov v patogenéze tohto ochorenia. Väčšina pacientov s NAFLD je obéznych, NAFLD sa vyskytuje ale aj u neobéznych jedincov a naopak, u obéznych pacientov môže absentovať. V klinickej praxi je transabdominálna ultrasonografia najvyužívanejšia skríningová zobrazovacia metóda NAFLD. Cieľ: Cieľom práce bolo posúdenie vplyvu najrizikovejšieho faktora, ktorým je detská obezita, na vznik a rozvoj NAFLD. Materiál a metodika: V priebehu ultrasonografického vyšetrenia sme kvantitatívne hodnotili stupeň stukovatenia pečene (vyjadreného hepatorenálnym indexom) a následne aj tuhosť pečeňového tkaniva (vyjadrenú indexom pečeňovej fibrózy) celkovo u 240 probandov v rôznych skupinách podľa vekových a hmotnostných kritérií. Výsledky ultrasonografického vyšetrenia sme následne korelovali s vybranými antropometrickými a laboratórnymi parametrami. Výsledky: Zdraví donosení novorodenci s normálnou pôrodnou hmotnosťou mali signifikantne nižšie hodnoty hepatorenálneho indexu (hepato renal index – HRI) a indexu pečeňovej fibrózy (liver fibrosis index – LFI) v porovnaní so zdravou kontrolnou pediatrickou skupinou probandov vo vekovom rozmedzí od 10. do ukončeného 18. roku života s normálnou hmotnosťou (p < 0,001). V kontrolnej skupine pubertálnych a adolescentných pacientov sme nezaznamenali vplyv pohlavia na zmeny ultrasonografických hodnôt HRI (p = 0,332) a LFI (p = 0,339). Fyziologické hodnoty hepatorenálneho indexu sa bez ohľadu na pohlavie pohybovali v kontrolnej pediatrickej skupine v rozmedzí 1,02–1,23 (10. až 90. percentil), resp. 1,02–1,26 (5. až 95. percentil). V skupine obéznych pediatrických pacientov vo vekovom rozmedzí od 10. do ukončeného 18. roku života bola preukázaná štatisticky signifikantne vyššia hodnota HRI aj LFI v porovnaní s kontrolnou pediatrickou skupinou s normálnou hmotnosťou. V skupine obéznych pacientov sa so zvyšujúcou hodnotou BMI znižovala elasticita pečene (p = 0,005; rs = 0,310), avšak stupeň steatózy sa nemenil (p = 0,357). U obéznych pacientov sa elasticita pečeňového tkaniva znižovala aj s rastúcim obvodom pása (p < 0,01). Záver: Výsledky našej práce poukazujú na významnú asociáciu obezity a NAFLD v pediatrickej populácii. Ultrasonografické metódy stanovovania hepatorenálneho indexu a elasticity pečene majú uplatnenie v diagnostike skorých stupňov pečeňového poškodenia u obéznych pubertálnych a adolescentných pacientov s rizikom vzniku NAFLD. Okrem skorého záchytu týchto zmien sme schopní neinvazívne a v reálnom čase hodnotiť dynamiku ochorenia a efekt podávanej terapie, čím sa zlepšuje kontrola nad ochorením.
Reference
1. Bodzsar EB, Zsakai A. Recent trends in childhood obesity and overweight in the transition countries of Eastern and Central Europe. Ann Hum Biol 2014; 41(3): 263–270. doi: 10.3109/03014460.2013.856473.
2. WHO. Obesity and overweight. 2021 [online]. Dostupné z: http: //who.int/mediacentre/factsheets/fs311/en/.
3. Cole TJ, Bellizzi MC, Flegal KM et al. Establishing a standard definition for child overweight and obesity worldwide: international survey. BMJ 2000; 320(7244): 1240–1243. doi: 10.1136/bmj.320.7244.1240.
4. Thomas DM, Bredlau C, Bosy-Westphal A et al. Relationships between body roundness with body fat and visceral adipose tissue emerging from a new geometrical model. Obesity (Silver Spring) 2013; 21(11): 2264–2271. doi: 10.1002/ oby.20408.
5. Webb M, Yeshua H, Zelber-Sagi S et al. Diag- nostic value of a computerized hepatorenal index for sonographic quantification of liver steatosis. AJR Am J Roentgenol 2009; 192(4): 909–914. doi: 10.2214/AJR.07.4016.
6. Kohli R, Sunduram S, Mouzaki M et al. Pediatric nonalcoholic fatty liver disease: a report from the expert committee on nonalcoholic fatty liver disease (ECON). J Pediatr 2016; 172: 9–13. doi: 10.1016/j.jpeds.2015.12.016.
7. Akcam M, Boyaci A, Pirgon O et al. Importance of the liver ultrasound scores in pubertal obese children with nonalcoholic fatty liver disease. Clin Imaging 2013; 37(3): 504–508. doi: 10.1016/j.clinimag.2012.07.011.
8. Hamaguchi M, Kojima T, Itoh Y et al. The severity of ultrasonographic findings in nonalcoholic fatty liver disease reflects the metabolic syndrome and visceral fat accumulation. Am J Gastroenterol 2007; 102(12): 2708–2715. doi: 10.1111/j.1572-0241.2007.01526.x.
9. Duarte MA, Silva GA. Hepatic steatosis in obese children and adolescents. J Pediatr 2011; 87(2): 150–156. doi: 10.2223/JPED.2065.
10. Vajro P, Lenta S, Socha P et al. Diagnosis of nonalcoholic fatty liver disease in children and adolescents: position paper of the ESPGHAN Hepatology Committee. J Pediatr Gastroenterol Nutr 2012; 54(5): 700–713. doi: 10.1097/MPG.0b013e318252a13f.
11. Manco M, Bedogni G, Marcellini M et al. Waist circumference correlates with liver fibrosis in children with non-alcoholic steatohepatitis. Gut 2008; 57(9): 1283–1287. doi: 10.1136/gut. 2007.142919.
12. Motamed N, Rabiee B, Hemasi GR et al. Body Roundness Index and Waist-to-Height Ratio are Strongly Associated with Non-Alcoholic Fatty Liver Disease: A Population-Based Study. Hepat Mon 2016; 16(9): e39575. doi: 10.5812/hepatmon.39575.
13. Michnová Z, Szépeová R, Havlíčeková Z et al. Prevalencia hepatopatie u adolescentov s diabetes mellitus 1. typu. Pediatria (Bratisl.) 2017; 12(2): 53.
14. Saadeh S, Younossi ZM, Remer EM et al. The utility of radiological imaging in nonalcoholic fatty liver disease. Gastroenterology 2002; 123(3): 745–750. doi: 10.1053/gast.2002.35354.
15. Shannon A, Alkhouri N, Carter-Kent C et al. Ultrasonographic quantitative estimation of hepatic steatosis in children With NAFLD. J Pediatr Gastroenterol Nutr 2011; 53(2): 190–195. doi: 10.1097/MPG.0b013e31821b4b61.
16. Borges VF, Diniz AL, Cotrim HP et al. Sonographic hepatorenal ratio: a noninvasive method to diagnose nonalcoholic steatosis. J Clin Ultrasound 2013; 41(1): 18–25. doi: 10.1002/jcu.21994.
17. Ayonrinde OT, Oddy WH, Adams LA et al. Infant nutrition and maternal obesity influence the risk of non-alcoholic fatty liver disease in adolescents. J Hepatol 2017; 67(3): 568–576. doi: 10.1016/j.jhep.2017.03.029.
18. Thompson AL. Intergenerational impact of maternal obesity and postnatal feeding practices on pediatric obesity. Nutr Rev 2013; 71(1): S55–S61. doi: 10.1111/nure.12054.
19. Ferraioli G, Calcaterra V, Lissandrin R et al. Noninvasive assessment of liver steatosis in children: the clinical value of controlled attenuation parameter. BMC Gastroenterol 2017; 17(1): 61. doi: 10.1186/s12876-017-0617-6.
20. Bailey SS, Youssfi M, Patel M et al. Shear-wave ultrasound elastography of the liver in normal-weight and obese children. Acta Radiologica 2017; 58(12): 1511–1518. doi: 10.1177/0284185117695668.
21. Cho Y, Tokuhara D, Morikawa H et al. Transient Elastography-Based Liver Profiles in a Hospital-Based Pediatric Population in Japan. PLoS One 2015; 10(9): e0137239. doi: 10.1371/journal.pone.0137239.
22. Fishbein MH, Miner M, Mogren C et al. The spectrum of fatty liver in obese children and the relationship of serum aminotransferases to severity of steatosis. J Pediatr Gastroenterol Nutr 2003; 36(1): 54–61. doi: 10.1097/00005176-200301000-00012.
23. Havlíčeková Z, Szépeová R, Zubríková L et al. Význam črevnej mikroflóry v etiopatogenéze nealkoholovej steatózy pečene. Čes-slov Pediat 2015; 70(1): 11–16.
24. Schwimmer JB, Deutsch R, Kahen T et al. Prevalence of fatty liver in children and adolescents. Pediatrics 2006; 118(4): 1388–1393. doi: 10.1542/peds.2006-1212.
25. Baker L, Farpour-Lambert JN, Nowicka P et al. Vyšetrenie dieťaťa s nadváhou/obezitou praktické rady pre primárnu lekársku starostlivosť: odporúčania od childhood obesity task force európskejobezitologickej spoločnosti (AESO). Pediatria (Bratisl.) 2012; 7(5): 229–234.
26. Račanská E. Vitamín D – hormón, ktorý nám chýba. Prakt. lekárn 2014; 4(2–3): 53–55.
27. Black LJ, Jacoby P, She Ping-Delfos WC et al. Low serum 25-hydroxyvitamin D concentracions associate with non-alcoholic fatty liver disease in adolescents independent of adiposity. J Gastroenterol Hepatol 2014; 29(6): 1215–1222. doi: 10.1111/jgh.12541.