Abstrakt
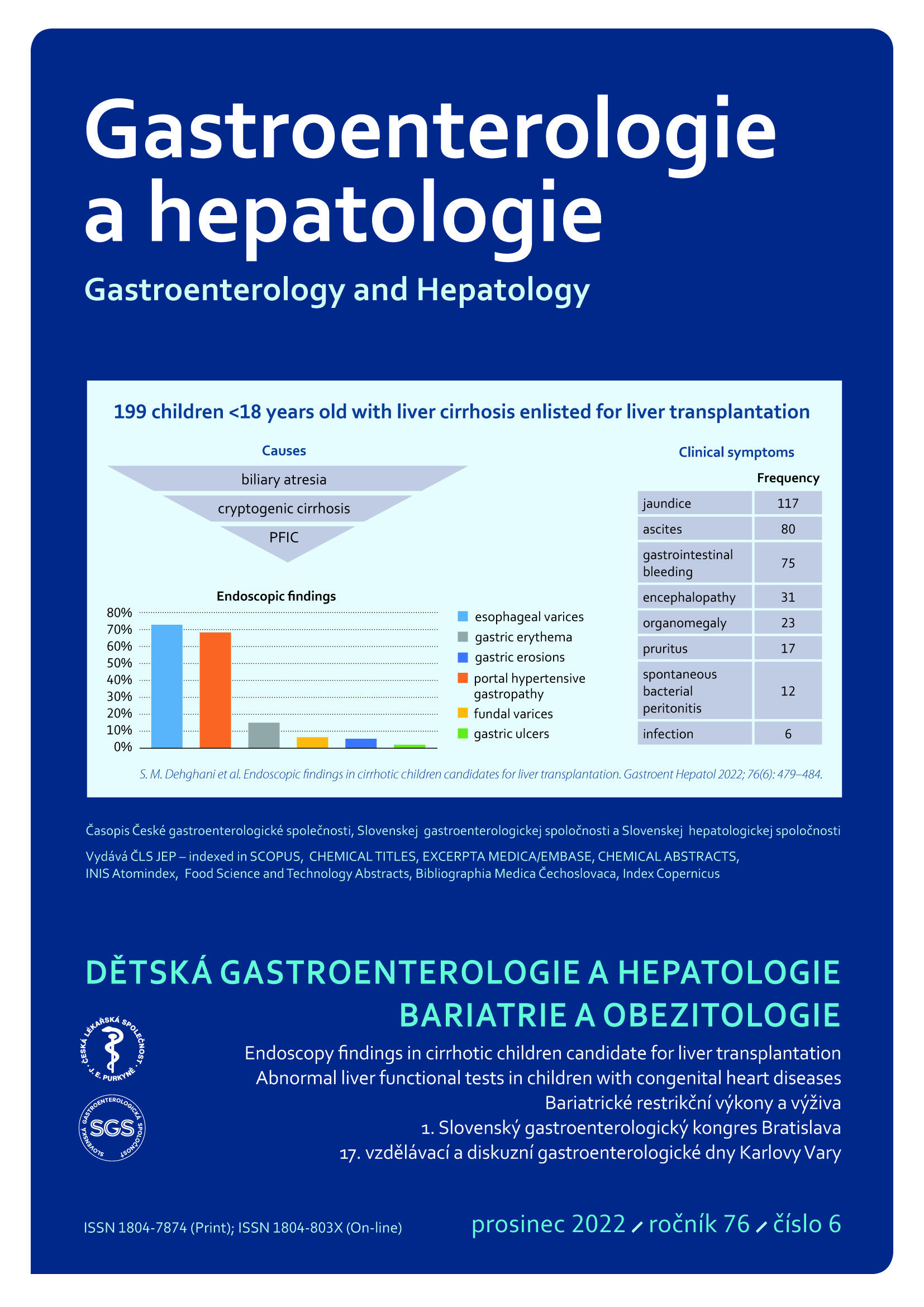
Východiska: Cirhóza je koncovým stadiem jaterního selhání, které se může rozvinout z akutního nebo chronického postižení jater. Pacientům hrozí fatální komplikace jako portální hypertenze nebo krvácení z jícnových varixů. Metody: Tato průřezová studie se zaobírala endoskopickými nálezy u dětských cirhotických pacientů (do 18 let) vybraných pro transplantaci jater. Účastníky byly děti přijaté mezi lety 2012 a 2017 do Shiraz Organ Transplantation Center of Nemazee Hospital, Shiraz University of Medical Sciences. Data jsme získali pomocí speciálně připraveného dotazníku. Výsledky: Celkem jsme zkoumali 199 dětí (z nich 49,2 % byli chlapci), které jsme podrobili endoskopii horní části zažívacího traktu. V průměru jim bylo 6,2 ± 4,7 let. Jejich nejčastějším klinickým projevem byl ikterus (58,8 %). Průměrná hodnota Child-Pugh skóre byla u pacientů 8,53 ± 2,34 a PELD/MELD skóre 14,85 ± 14,93. Z celkového počtu 199 pacientů se jícnové varixy prokázaly u 145 (72,8 %), z nichž 39 (19,7 %) dosáhlo stupně 1, 53 (26,8 %) stupně 2, 42 (21,2 %) stupně 3 a 11 (5,6 %) stupně 4 dle závažnosti. Další projevy se objevily u 31,1 % – portální hypertenzní gastropatie (PHG), 15,1 % – erytém žaludeční sliznice, 6,5 % – fundální varixy, 5,5 % – eroze žaludeční sliznice a 2 % – žaludeční vřed. Nebyla pozorována žádná souvislost mezi výskytem jícnových nebo žaludečních vředů a skórem Child-Pugh nebo PELD/MELD, klinickými projevy, laboratorními výsledky nebo jinými probíhajícími onemocněními. Závěr: Nejčastější příčinou jaterní cirhózy u našich pacientů byla biliární atrezie a nejčastějším endoskopickým nálezem u nich byly jícnové varixy.
Reference
1. D‘Amico G, Garcia-Tsao G, Pagliaro L. Natural history and prognostic indicators of survival in cirrhosis: a systematic review of 118 studies. J Hepatol 2006; 44(1): 217–231. doi: 10.1016/j.jhep.2005.10.013.
2. Garcia‐Tsao G, Friedman S, Iredale J et al. Now there are many (stages) where before there was one: In search of a pathophysiological classification of cirrhosis. Hepatology 2010; 51(4): 1445–1449. doi: 10.1002/hep.23478.
3. Ginés P, Quintero E, Arroyo V et al. Compensated cirrhosis: natural history and prognostic factors. Hepatology 1987; 7(1): 122–128. doi: 10.1002/hep.1840070124.
4. D’Amico G. Esophageal varices: from appearance to rupture; natural history and prognostic indicators. Portal hypertension in the 21st Century. Springer 2004; 147–154. doi: 10.1007/ 978-94-007-1042-9_17.
5. Owensby S, Taylor K, Wilkins T. Diagnosis and management of upper gastrointestinal bleeding in children. J Am Board Fam Med 2015; 28(1): 134–145. doi: 10.3122/jabfm.2015.01.140 153.
6. D‘Amico G, Garcia‐Tsao G, Cales P et al. Session 2 – Diagnosis of Portal Hypertension: How and When. In: Portal Hypertension III: Pro- ceedings of the Third Baverno International Consensus Workshop on Definitions, Methodology and Therapeutic Strategies; 2001: Wiley Online Library. doi: 10.1002/97804707601 54.ch4.
7. Gado AS, Ebeid BA, Abdelmohsen AM et al. Clinical outcome of acute upper gastrointestinal hemorrhage among patients admitted to a government hospital in Egypt. Saudi J Gastroenterol 2012; 18(1): 34–39. doi: 10.4103/13 19-3767.91737.
8. Carbonell N, Pauwels A, Serfaty L et al. Improved survival after variceal bleeding in patients with cirrhosis over the past two decades. Hepatology 2004; 40(3): 652–659. doi: 10.1002/hep.20339.
9. Toubia N, Sanyal AJ. Portal hypertension and variceal hemorrhage. Med Clin North Am 2008; 92(3): 551–574, viii. doi: 10.1016/ j.mcna.2007.12.003.
10. Merli M, Nicolini G, Angeloni S et al. Incidence and natural history of small esophageal varices in cirrhotic patients. J Hepatol 2003; 38(3): 266–272. doi: 10.1016/s0168-8278(02)00420-8.
11. Garcia‐Tsao G, Bosch J, Groszmann RJ. Portal hypertension and variceal bleeding – Unresolved issues. Summary of an American Association for the study of liver diseases and European Association for the study of the liver single‐topic conference. Hepatology 2008; 47(5): 1764–1772. doi: 10.1002/hep.22273.
12. Pinto RB, Schneider AC, da Silveira TR. Cirrhosis in children and adolescents: An overview. World J Hepatol 2015; 7(3): 392–405. doi: 10.4254/wjh.v7.i3.392.
13. Chaabouni M, Bahloul S, Ben Romdhane W et al. Epidemiological, etiological and evolutionary aspects of children cirrhosis in a developing country: experience of the pediatric department of SFAX University hospital, Tunisia. Tunis Med 2007; 85(9): 738–743.
14. Dehghani SM, Shahramian I, Bazi A et al. Evaluation of Underlying Liver Disease and Its Severity in Children Referred for Liver Transplant: a Single-Center Report From Nemazee Hospital of Shiraz. Exp Clin Transplant 2020; 18(7): 803–807. doi: 10.6002/ect.2018.0047.
15. Dehghani SM, Imanieh MH, Haghighat M et al. Etiology and complications of liver cirrhosis in children: report of a single center from southern iran. Middle East J Dig Dis: 2013; 5(1): 41–46.
16. Ng NB, Karthik SV, Aw MM et al. Endoscopic Evaluation in Children With End-Stage Liver Disease-Associated Portal Hypertension Awaiting Liver Transplant. J Pediatr Gastroenterol Nutr 2016; 63(3): 365–369. doi: 10.1097/ MPG.0000000000001160.
17. Bonnet N, Paul J, Helleputte T et al. Novel insights into the assessment of risk of upper gastrointestinal bleeding in decompensated cirrhotic children. Pediatr Transplant 2019; 23(4): e13390. doi: 10.1111/petr.13390.
18. Tumgor G, Arikan C, Yuksekkaya HA et al. Childhood cirrhosis, hepatopulmonary syndrome and liver transplantation. Pediatr Transplant 2008; 12(3): 353–357. doi: 10.1111/j.13 99-3046. 2007.00807.x.
19. Gunda DW, Kilonzo SB, Mamballah Z et al. The magnitude and correlates of esophageal Varices among newly diagnosed cirrhotic patients undergoing screening fibre optic endoscope before incident bleeding in North-Western Tanzania; a cross-sectional study. BMC Gastroenterol 2019; 19(1): 203. doi: 10.1186/s12876-019-1123-9.
20. Saleh ZM, Solano QP, Louissaint J et al. The incidence and outcome of postoperative hepatic encephalopathy in patients with cirrhosis. United European Gastroenterol 2021; 9(6): 672–680. doi: 10.1002/ueg2.12104.
21. Fujiyama S, Akuta N, Sezaki H et al. Mortality rates and risk factors in 1412 Japanese patients with decompensated hepatitis C virus-related cirrhosis: a retrospective long-term cohort study. BMC Gastroenterol 2021; 21(1): 189. doi: 10.1186/s12876-021-01770-0.
22. Xu SH, Wu F, Guo LH et al. Liver fibrosis index-based nomograms for identifying esophageal varices in patients with chronic hepatitis B related cirrhosis. World J Gastroenterol 2020; 26(45): 7204–7221. doi: 10.3748/wjg.v26.i45. 7204.
23. Xu X, Jin Y, Lin Y et al. Multimodal Ultrasound Model Based on the Left Gastric Vein in B-Viral Cirrhosis: Noninvasive Prediction of Esophageal Varices. Clin Transl gastroenterol 2020; 11(1): e00262. doi: 10.14309/ctg.0000000000000262.
24. Fierbinteanu-Braticevici C, Tribus L, Peagu R et al. Spleen Stiffness as Predictor of Esophageal Varices in Cirrhosis of Different Etiologies. Sci Rep 2019; 9(1): 16190. doi: 10.1038/s4159 8-019-52407-y.
25. Yang LB, Xu JY, Tantai XX et al. Non-invasive prediction model for high-risk esophageal varices in the Chinese population. World J Gastroenterol 2020; 26(21): 2839–2851. doi: 10.3748/wjg.v26.i21.2839.
26. Ekmen N, Cifci S. Evaluation of the Relationship Between Pulmonary Artery Hypertension and Esophageal Varices Bleeding in Transplantation Candidates. Cureus 2021; 13(2): e13355. doi: 10.7759/cureus.13355.
27. Takehara T, Sakamori R. Remaining challenges for the noninvasive diagnosis of esophageal varices in liver cirrhosis. Esophagus 2020; 17(1): 19–24. doi: 10.1007/s10388-019-00699-4.
28. Zhou H, Long J, Hu H et al. Liver stiffness and serum markers for excluding high-risk varices in patients who do not meet Baveno VI criteria. World J Gastroenterol 2019; 25(35): 5323–5333. doi: 10.3748/wjg.v25.i35.5323.
29. Qi X, Li Y, Wang R et al. Liaoning Score for Prediction of Esophageal Varices in Cirrhotic Patients Who Had Never Undergone Endoscopy: A Multicenter Cross-Sectional Study in Liaoning Province, China. Adv Ther 2019; 36(8): 2167–2178. doi: 10.1007/s12325-019-00967-w.
30. Simbrunner B, Beer A, Wöran K et al. Portal hypertensive gastropathy is associated with iron deficiency anemia. Wien Klin Wochenschr 2020; 132(1–2): 1–11. doi: 10.1007/s00 508-019-01593-w.
31. Purbey BK, Gurung RB, Panday R et al. The Etiology of Upper Gastrointestinal Bleeding in Patients with Liver Cirrhosis in Dhulikhel Hospital. Kathmandu Univ Med J (KUMJ) 2017; 15(60): 292–295.
32. Mandhwani R, Hanif FM, Ul Haque MM et al. Noninvasive Clinical Predictors of Portal Hypertensive Gastropathy in Patients with Liver Cirrhosis. J Transl Int Med 2017; 5(3): 169–173. doi: 10.1515/jtim-2017-0025.
33. Singh S, Bhamre R, Shetty N et al. Correlation of endoscopic findings with Doppler ultrasound in portal hypertension in children. Clin Exp Hepatol 2021; 7(2): 191–195. doi: 10.5114/ceh.2021.106509.
34. Ng NBH, Karthik SV, Aw MM et al. Endoscopic Evaluation in Children With End-Stage Liver Disease-Associated Portal Hypertension Awaiting Liver Transplant. J Pediatr Gastroenterol Nutr 2016; 63(3): 365–369. doi: 10.1097/MPG.0000000000001160.
35. Min YW, Bae SY, Gwak GY et al. A clinical predictor of varices and portal hypertensive gastropathy in patients with chronic liver disease. Clin Mol Hepatol 2012; 18(2): 178–184. doi: 10.3350/cmh.2012.18.2.178.
36. Fontana RJ, Sanyal AJ, Ghany MG et al. Development and progression of portal hypertensive gastropathy in patients with chronic hepatitis C. Am J Gastroenterol 2011; 106(5): 884–893. doi: 10.1038/ajg.2010.456.
37. Sathar SA, Kunnathuparambil SG, Sreesh S et al. Helicobacter pylori infection in patients with liver cirrhosis: prevalence and association with portal hypertensive gastropathy. Ann Gastroenterol 2014; 27(1): 48–52.
38. Kunihara S, Oka S, Tanaka S et al. Predictive Factors of Portal Hypertensive Enteropathy Exacerbation in Patients with Liver Cirrhosis: A Capsule Endoscopy Study Digestion 2018; 98(1): 33–40. doi: 10.1159/000486666.
39. Samiullah S, Memon MS, Memon HG et al. Secondary gastric varices in hepatic cirrhosis. J Coll Physicians Surg Pak 2011; 21(10): 593–596. doi: 10.2011/JCPSP.593596.
40. De Faria AA, Dias CAF, Dias Moetzsohn L et al. Feasibility of transnasal endoscopy in screening for esophageal and gastric varices in patients with chronic liver disease. Endosc Int Open 2017; 5(7): 646–651. doi: 10.1055/s-0043-107781.
41. Butt Z, Ali Shah SM, Afzal M et al. Frequency of different types of gastric varices in patients with cirrhosis due to chronic hepatitis C. J Pak Med Assoc 2016; 66(11): 1462–1465.
42. Petrisor A, Stanescu AMA, Papacocea IR et al. Non-invasive laboratory, imaging and elastography markers in predicting varices with high risk of bleeding in cirrhotic patients. Rom J Intern Med 2021; 59(2): 194–200. doi: 10.2478/rjim-2021-0001.
43. Lu Z, Sun X, Han J et al. Characteristics of peptic ulcer bleeding in cirrhotic patients with esophageal and gastric varices. Sci Rep 2020; 10(1): 20068. doi: 10.1038/s41598-020-76530-3.
44. Lesmana CRA, Kalista KF, Sandra S et al. Clinical significance of isolated gastric varices in liver cirrhotic patients: A single-referral-centre retrospective cohort study. JGH Open 2019; 4(3): 511–518. doi: 10.1002/jgh3.12292.