Biosimilar infliximab in anti-TNF-naïve IBD patients – 1-year clinical follow-up
Martin Kolář1, Dana Ďuricová Orcid.org 1, Martin Bortlík Orcid.org 1,2,3, Veronika Hrubá1, Naděžda Machková Orcid.org 1, Katarína Mitrová Orcid.org 1,4, Karin Malíčková Orcid.org 1,5, Martin Lukáš Orcid.org 1, Milan Lukáš Orcid.org 1
+ Affiliation
Summary
Background: Biosimilar infliximab (IFX) has been approved for the treatment of inflammatory bowel disease (IBD) in the European Union since September 2013. The approval process included extrapolation of clinical data from other indications, namely, rheumatoid arthritis and ankylosing spondylitis. Data from clinical practice are therefore desirable to confirm the efficacy and safety of biosimilar IFX in the IBD population. Evidence of the long-term efficiency and safety of maintenance treatment with biosimilar IFX in patients with IBD is sparse. Methods: Data from consecutive patients with Crohn’s disease (CD) and ulcerative colitis (UC) who were started on biosimilar IFX between January 2015 and May 2016 at our center were analyzed. Patients were assessed as non-responders, partial responders, or complete responders based on clinical, endoscopic, and laboratory parameters. Besides clinical and endoscopic evaluation, C reactive protein levels, fecal calprotectin levels, blood counts, IFX trough levels, and anti-IFX antibody levels were measured. All adverse events were recorded. Final analysis was performed at week 54. Results: One hundred and forty IBD patients (CD, 107; UC, 33) were included in the analysis. In total, 94% of CD patients and 82% of UC patients responded to induction therapy with biosimilar IFX. At week 54, the response rates were 87% in CD patients and 60% in UC patients, and 47% and 36% of CD and UC patients, respectively, were in remission. Fifty-two percent of UC patients demonstrated mucosal healing at week 14. Perianal disease improved in 96% of CD patients at week 54, including 59% with complete cessation of drainage. A marked steroid-sparing effect was observed in all patients. Therapy was continued in 83% of patients at the end of the follow-up (CD, 89%; UC, 64%). Pharmacokinetic properties, immunogenicity, and the frequencies and characteristics of adverse events were similar to those previously observed during treatment with the original IFX. Conclusion: Biosimilar IFX seems to be effective and safe in IBD patients; it achieves similar results as the original preparation with no additional safety issues.
Keywords
infliximab, inflammatory bowel disease, biosimilar pharmaceuticalsIntroduction
Biologic therapy is an effective, but also expensive treatment of different autoimmune diseases, including inflammatory bowel diseases (IBD). Biosimilar is a term referring to another version of an already licensed biologic drug with no meaningful differences from the reference product in terms of quality, physicochemical properties, biologic activity, safety or efficacy. Introduction of the first biosimilar infliximab (IFX) (CT-P13) was aimed at reducing the health care cost burden and to increase the number of treated individuals with similar treatment results. CT-P13 has been given authorisation in the European Union in September 2013 and the Food and Drug Administration authorised the product for use on the US market in April 2016. More recently, in May 2016, the European Medicines Agency (EMA) authorised for use the second biosimilar IFX named Flixabi®.
After thorough initial physicochemical, pharmacodynamic and pharmacokinetic comparative studies, two randomised controlled studies showed that CT-P13 has comparable safety and efficacy to the originator product in patients with ankylosing spondylitis and rheumatoid arthritis [1–3]. Subsequently, these findings led the EMA to extrapolation of the clinical data to other indications of the reference product, including IBD, even though previous studies in this specific patient population were not available. Since then, the use of biosimilar IFX has grown extensively, and several reports have confirmed comparable efficacy and safety with the originator product in anti-TNF-a naïve patients with IBD [4–8].
Even though the information in anti-TNF-a naïve patients with IBD are becoming quite solid, there is still a lack of sufficient data on long-term results in IBD patients on maintenance treatment with biosimilar IFX. Our aim was thus to evaluate the efficiency and safety of biosimilar IFX one year after routine usage in clinical practice in anti-TNF-a naïve IBD patients.
Patients and Methods
Consecutive patients with Crohn’s disease (CD) and ulcerative colitis (UC) naïve to anti-TNF-a in whom IFX therapy was indicated during a period from January 2015 to May 2016 were included.
Patients were followed in regular intervals coincident with infusion applications. At each visit a blood sample was taken for analysis of blood count, biochemistry and IFX pharmacokinetics (trough levels (TL) and anti-drug antibodies (ATI)) and a stool sample was collected for measurement of faecal calprotectin (FC). Intervals between infusions as well as dose were determined by a treating physician according to the patients’ needs. Response of both CD and UC patients naïve to biosimilar IFX was assessed retrospectively at weeks 14 (W14) and 54 (W54) based on clinical, endoscopic and biologic markers. In addition, endoscopic response in UC patients measured by change in Mayo score from baseline was assessed at W14. Adverse events were registered at each visit.
Measurement of pharmacokinetic parameters
IFX TLs and ATIs of biosimilar IFX were measured by quantitative enzyme-linked immunosorbent assay (ELISA) kit SHIKARI Q-Inflixi and Q-ATI, respectively, manufactured by Matriks Biotek, Turkey. The lowest detectable limit of Q-Inflixi kit was 0.03 µg/mL. The presence of ATIs was determined qualitatively by comparing the share of optical density of a sample at 450 nm and the mean optical density at 450 nm of negative controls with an index of 3. If the share was ≥ 3 the sample was positive for ATIs.
Statistical analysis
Standard descriptive statistical analyses were performed, including frequency distributions for categorical data and calculation of mean and standard deviation and/or range for continuous variables. The differences of continuous variables between baseline and W54 were analysed using the Mann- -Whitney test. Using survival analysis, Kaplan-Meier curve was plotted to depict the proportion of patients remaining on the therapy. The IFX TL cut-off value predicting long-term response to therapy was calculated by receiver operating characteristic (ROC) in conjunction with Youden’s J statistic. Evaluation of categorical variables difference was performed by Fisher’s exact test. The statistical tests were performed using GraphPad Prism (version 6.05). A p-value < 0.05 was considered statistically significant.
Results
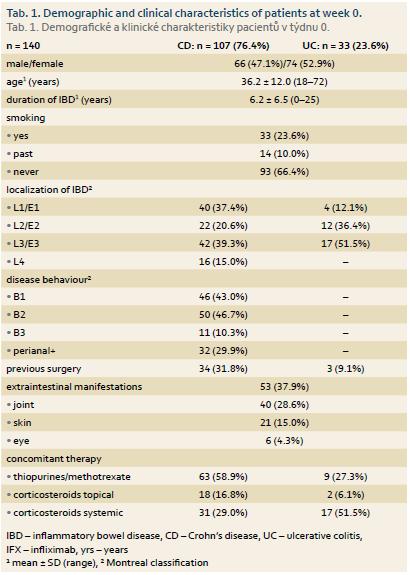
IFX treatment was newly initiated in 140 patients, 107 with CD and 33 with UC. Mean disease duration in these patients was 6.2 ± 6.5 years. Concomitant immunosuppression was present in 63 (58.9%) patients with CD and 9 (27.3%) with UC, 31 (29.0%) CD patients and 17 (51.5%) UC patients were on systemic corticotherapy and 18 (16.8%) and 2 (6.1%) patients had topical corticosteroids, respectively. Perianal disease was present at the time of treatment initiation or in disease history in 32 (29.9%) of CD patients and mean Mayo score in UC patients was 2.7. Baseline mean ± SD (range) C reactive protein (CRP) values in naïve patients were 13.3 ± 19.5 mg/L (0.2–145.9) in CD patients and 9.6 ± 17.1 mg/L (0.4–81.2) in UC patients. W0 FC levels were 558 ± 357 μg/g (30–1,000) and 672 ± 425 mg/L (30–1,000), respectively. The demographic and clinical characteristics of IFX naïve patients are outlined in Tab. 1.
Efficiency
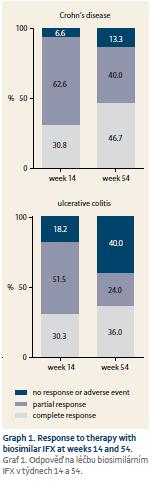
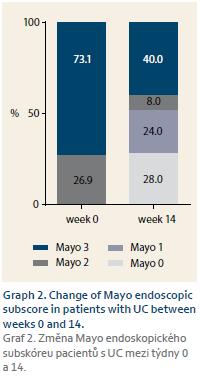
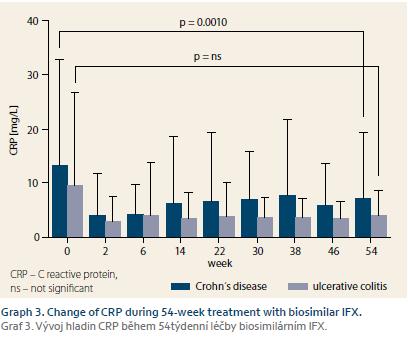
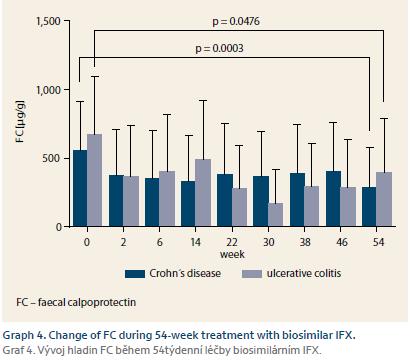
Response of all 140 naïve patients was evaluated at W14. Complete response to therapy was observed in 33 (30.8%) CD and 10 (30.3%) UC patients. Partially responded 67 (62.6%) CD and 17 (51.5%) UC patients, and no response occurred in 7 (6.5%) CD and 6 (18.2%) UC patients. Furthermore, response of 100 patients who reached W54 or who terminated the therapy early was evaluated. Of these patients, 35 (46.7%) CD and nine (36.0%) UC patients completely responded to the therapy. Partial response occurred in 30 (40.0%) CD and 6 (24.0%) UC patients, and no response was observed in 10 (13.3%) and 10 (40.0%) patients, respectively (Graph 1). Mean Mayo score in UC patients dropped from 2.7 at W0 to 1.6 at W16 (p = 0.0002). Mucosal healing (Mayo endoscopic subscore 0 or 1) was achieved in 13 (52.0%) of UC patients with Mayo endoscopic subscore of 3 or 2 at baseline (Graph 2). All 22 patients with active perianal disease were adequately drained and observed by surgeons. Of these patients, the disease was completely resolved in 13 (59.1%) patients by W54, 8 (36.4%) patients experienced improvement of their condition, and 1 (4.5%) patient did not respond to biosimilar IFX treatment, however this patient had to be transferred to adalimumab early after the therapy initiation due to SLE-like symptoms. Comparing W0 and W54, a significant drop in CRP level was observed in CD patients (13.3 ± 19.5 mg/L vs. 7.1 ± 12.2 mg/L; p = 0.0010). In UC patients, only numerical decrease was present not reaching statistical significance (9.6 ± 17.1 mg/L vs. 4.0 ± 4.7 mg/L) (Graph 3). Similarly, the mean value of FC differed significantly between baseline and the time of evaluation in both CD and UC (558 ± 357 µg/g vs. 290 ± 288 µg/g; p = 0.0003 and 672 ± 425 µg/g vs. 396 ± 392 µg/g; p = 0.0476) (Graph 4).
The proportion of patients on systemic corticotherapy dropped from 29.0% at W0 to 1.6% at W54 in CD patients and from 51.5% to 6.3% in UC patients. Of 16.8% CD and 6.1% UC patients receiving topical corticosteroids at baseline, none had the topical steroids at W54. Concomitant immunosuppression remained stable during the time of observation, changing from 58.9% at W0 to 60.3% at W54 in patients with CD and from 27.3% to 31.3% of patients with UC.
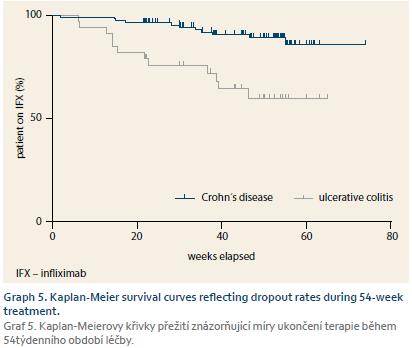
In total, therapy with biosimilar IFX was stopped in 24 (17.1%) patients – 12 (11.2%) CD and 12 (36.4%) UC by the end of the follow-up. Estimated failure-free survival at 54 weeks of therapy is 89.2% for CD and 59.7% for UC (Graph 5). Two CD patients ended treatment due to infusion reaction and five for other adverse events. Three patients with CD stopped anti-TNF after undergoing IC resection and in two patients either follow-up was lost or they voluntarily quitted treatment. Of 12 UC patients, three were primary non-responders and seven patients failed during the maintenance phase. Two patients experienced adverse reactions that led to treatment stop.
Treatment optimisation
During the follow-up 36 (25.7%) patients in total underwent some treatment intensifying optimisation – dose increase, interval shortening or both. Six (18.2%) UC patients initiated the therapy with increased dose (10 mg/kg) as a rescue therapy. Of these, two were primary non-responders and one lost response in a maintenance phase. Three patients continued treatment with regular dosing. At W54 17 (27.0%) CD and 9 (14.3%) UC patients had an intensified treatment regimen.
Safety
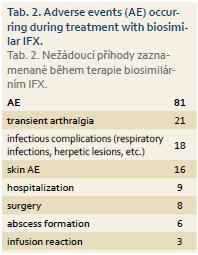
Three patients in whole cohort experienced infusion reaction. In total, 81 adverse events (AEs) were registered in 72 (51.4%) patients during the follow-up which represents 71.9 AEs per 100 patient-years. The frequency and type of adverse events were similar to those observed during the treatment with original IFX (Tab. 2) [9]. Seven patients in total discontinued biosimilar IFX due to adverse events other than infusion reaction – five patients discontinued therapy due to SLE-like reactions, severe arthralgia or skin rashes, one patient had severe infectious complications with formation of several abscesses, and the last patient suffered from palmoplantar pustulosis.
Pharmacokinetics and immunogenicity
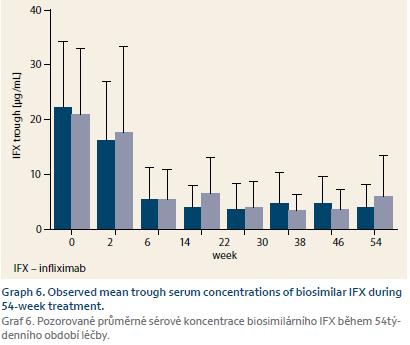
Pharmacokinetic properties of biosimilar IFX were comparable to the original preparation. Mean IFX TLs were similar for both diagnoses and for whole cohort at W2, W6, and W14 were 21.9 ± 12.0 μg/mL, 16.5 ± 12.0 μg/mL, and 5.4 ± 5.7 μg/mL, respectively. IFX TL at W54 was 4.4 ± 5.1 μg/mL (Graph 6). ATI positivity was present in 2.4% of patients at W14 and in 12.1% at W54. During the follow-up, ATI positivity occurred in 26 (18.6%) patients, however in 13 (50%) of these the antibodies were only transient.
Infliximab serum concentrations’ predictive role
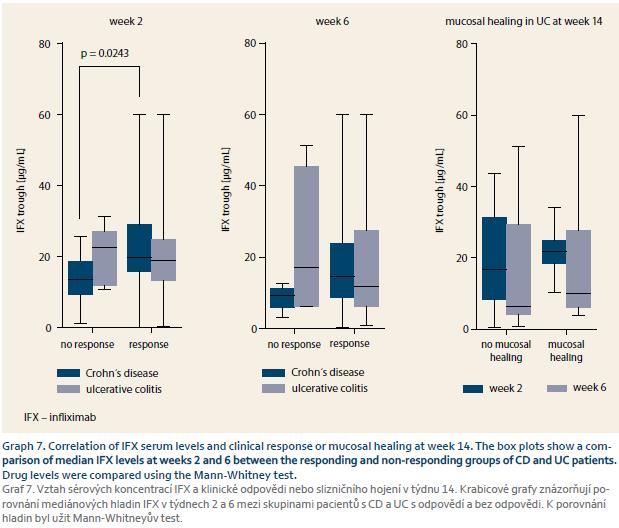
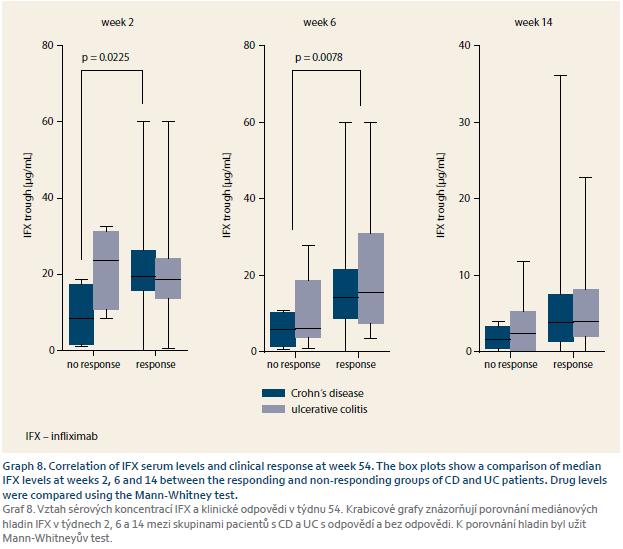
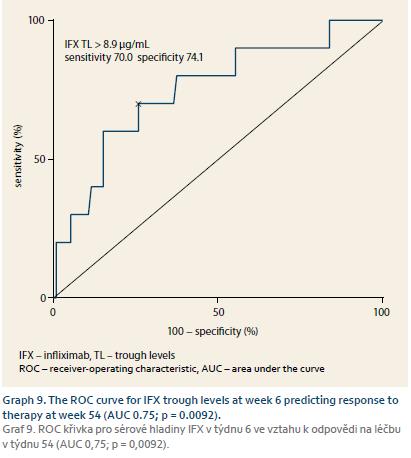
IFX serum concentrations were available in every patient and the predictive role of early trough levels for long-term response could have thus been estimated. IFX TLs of W2 were predictive for response in both W14 and W54 in CD. TLs at W6 were predictive for response at W54 for both CD and UC and were thus considered for cut--off value estimation where IFX TL of at least 8.9 μg/mL at W6 was best predictive of response at W54 with a sensitivity of 70.0% and specificity of 74.1% (Graph 7–9).
Discussion
In our observation, the patient cohort included anti-TNF-a naïve patients only. In total, 94% of CD and 82% of UC patients responded to induction therapy (W14) with biosimilar IFX. Reported by Schnitzler et al., the initial response to IFX therapy in CD on the original IFX was 89.1% [10]. Response rates at W8 achieved in 5 mg/kg groups of UC patients in ACT-1 and ACT-2 studies were 69% and 65%, respectively. [11]. At W54 the response rate in our cohort was 87% in CD and 60% in UC and 47% and 36% of patients were in remission, respectively. Previously in the analysis of Accent I trial response rate to IFX in CD at W54 was 63% and 41% of patients were in remission [12]. ACT-1 clinical response rate after 54 weeks was reported to be 46% and remission was reached by 35% of UC patients [11]. More than half of our UC patients experienced mucosal healing at W14 and improvement of perianal disease occurred in 96% of CD patients at W54, including 59.1% with complete cessation of drainage. In a study by Present et al. complete response of perianal fistulae to induction phase with original IFX occurred in 55% of patients [13]. Clear steroid-sparing effect was observed in all patients when steroids could have been almost completely tapered out during the follow-up period.
The therapy was continued in 83% of patients at the end of the follow-up (89% CD and 64% UC). Our group has previously published results from a cohort on original IFX where sustained response at one year was estimated to be 73%, however failed cases there also included patients with newly started corticosteroids or immunomodulators or their dose increase, as well as patients undergoing surgery [14]. On the other hand, a recently published study by Billiet et al. that includes patients on IFX from a 20-year timespan of clinical experience estimates 1 year failure-free survival of 94% [15].
Type and frequency of adverse events were comparable to original preparation as reported by cohort study with original IFX by Fidder et al. which focused on long-term safety [9].
Pharmacokinetic properties during maintenance phase of treatment remain stable and at W54 the mean IFX TL was 4.4 ± 5.1 μg/mL, being similar to value observed in our previous cohort of patients on original IFX where median TL measured at W14–W22 was 4.04 μg/mL. Similarly, cumulative ATI positivity occurred in 26 (18.6%) patients on biosimilar IFX during the follow-up compared to 17% recorded in Bortlik et al. [14].
It has already been reported that higher IFX TLs during induction are in association with long-term clinical response, mucosal healing and perianal fistulae closure [15–19]. We evaluated the predictive role of early TLs on long-term response in our cohort of IBD patients on biosimilar IFX. Trough level of at least 8.9 μg/mL at W6 was best predictive of response at W54 which corresponds well with the results of previous studies on original IFX. Our findings thus suggest that the same association applies also to the biosimilar IFX.
Efficiency and safety of biosimilar IFX in IBD have already also been presented in previous studies. In a work by Jahnsen et al. from Norway [8], 79% of CD and 56% of UC patients achieved remission at W14. No unexpected adverse events were reported during the study. In a Hungarian multicentric nationwide study [5], efficiency in the induction of clinical remission and response in both CD and UC was described at W14 and W30. This study also included patients with previous IFX exposure in whom decreased response rates and higher probability of developing allergic react- ions were observed. The biosimilar IFX has also been shown to be efficient in mucosal healing that was achieved in two-thirds of UC patients by the end of the induction period [6].
Despite similar clinical efficiency and safety of biosimilar and original IFX, even small differences in molecular structure might be responsible for different immunogenicity. Recent works have supported the immunogenic similarity between original and biosimilar IFX in IBD by proving that antibodies to original IFX similarly recognised and cross-reacted with the biosimilar IFX [20,21]. The usage of SHIKARI Q-Inflixi assay for the detection of biosimilar IFX concentration has been previously backed up by a study evaluating technical performance and confirming robustness and suitability of the assay for detection of biosimilar IFX serum levels [22].
According to our results and reports currently available, there is no negative or harmful signal about biosimilar IFX in anti-TNF-a naïve patients and its efficiency and safety seems to be comparable to the original preparation. The most important benefit from the introduction of biosimilars is a reduction of therapy costs. This is very important mainly in patients suffering from chronic diseases such as IBD who require long-standing treatment. An economic model for the introduction of biosimilar IFX in five West European countries showed potential considerable cost-saving effects even within one year of therapy [23]. Similarly, a 3-year budget impact analysis in countries from Central and Eastern Europe reported substantial cost savings by using biosimilar IFX [24]. This decrease in price might increase accessibility of the therapy for more patients.
Our observation has several limitations. Most importantly, we were not able to include a control group of naïve patients starting on the original product in order to perform a direct comparison between originator and biosimilar IFX in a randomised set of patients. Also, the number of patients enrolled into our cohorts is limited, especially those with UC.
In conclusion, biosimilar IFX in IBD patients seems to be effective and safe, gaining similar treatment results with no additional safety issues in comparison to the original preparation. However, there is a need for further mainly randomised controlled studies with larger patient populations and longer follow-up periods.
The authors declare he they have no potential conflicts of interest concerning drugs, products, or services used in the study.
Autoři deklarují, že v souvislosti s předmětem studie nemají žádné komerční zájmy.
The Editorial Board declares that the manuscript met the ICMJE „uniform requirements“ for biomedical papers.
Redakční rada potvrzuje, že rukopis práce splnil ICMJE kritéria pro publikace zasílané do biomedicínských časopisů.
Submitted/Doručeno: 23. 10. 2016
Accepted/Přijato: 23. 11. 2016
Prof. Milan Lukas, MD, PhD
IBD Clinical and Research Centre, ISCARE I.V.F. a. s.
Jankovcova 1569/2c
170 04 Prague 7
Czech Republic
milan.lukas@email.cz
To read this article in full, please register for free on this website.
Benefits for subscribers
Benefits for logged users
Literature
1. Park W, Yoo DH, Jaworski J et al. Comparable long-term efficacy, as assessed by patient-reported outcomes, safety and pharmacokinetics, of CT-P13 and reference infliximab in patients with ankylosing spondylitis: 54-week results from the randomized, parallel-group PLANETAS study. Arthritis Res Ther 2016; 18: 25. doi: 10.1186/s13075-016-0930-4.
2. Yoo DH, Racewicz A, Brzezicki J et al. A phase III randomized study to evaluate the efficacy and safety of CT-P13 compared with reference infliximab in patients with active rheumatoid arthritis: 54-week results from the PLANETRA study. Arthritis Res Ther 2016; 18: 82. doi: 10.1186/s13075-016-0981-6.
3. Choe JY, Prodanovic N, Niebrzydowski J et al. A randomised, double-blind, phase III study comparing SB2, an infliximab biosimilar, to the infliximab reference product Remicade in patients with moderate to severe rheumatoid arthritis despite methotrexate therapy. Ann Rheum Dis 2015. In press. doi: 10.1136/ annrheumdis-2015-207764.
4. Keil R, Wasserbauer M, Zádorová Z et al. Clinical monitoring: infliximab biosimilar CT-P13 in the treatment of Crohn’s dis- ease and ulcerative colitis. Scand J Gastroenterol 2016; 51 (9): 1062–1068. doi: 10.3109/00365521.2016.1149883.
5. Gecse KB, Lovász BD, Farkas K et al. Efficacy and safety of the biosimilar infliximab CT-P13 treatment in inflammatory bowel diseases: a prospective, multicentre, nationwide cohort. J Crohns Colitis 2016; 10 (2): 133–140. doi: 10.1093/ ecco-jcc/jjv220.
6. Farkas K, Rutka M, Golovics PA et al. Efficacy of infliximab biosimilar CT-P13 induction therapy on mucosal healing in ulcerative colitis. J Crohns Colitis 2016; 10 (11): 1273–1278.
7. Jung YS, Park DI, Kim YH et al. Efficacy and safety of CT-P13, a biosimilar of infliximab, in patients with inflammatory bowel disease: a retrospective multicenter study. J Gastroenterol Hepatol 2015; 30 (12): 1705–1712. doi: 10.1111/jgh.12997.
8. Jahnsen J, Detlie TE, Vatn S et al. Biosimilar infliximab (CT-P13) in the treatment of inflammatory bowel disease: a Norwegian observational study. Expert Rev Gastroenterol Hepatol 2015; 9 (Suppl 1): 45–52. doi: 10.1586/17474124.2015.1091308.
9. Fidder H, Schnitzler F, Ferrante M et al. Long-term safety of infliximab for the treatment of inflammatory bowel disease: a single-centre cohort study. Gut 2009; 58 (4): 501–508. doi: 10.1136/gut. 2008.163642.
10. Schnitzler F, Fidder H, Ferrante M et al. Long-term outcome of treatment with infliximab in 614 patients with Crohn’s disease: results from a single-centre cohort. Gut 2009; 58 (4): 492–500. doi: 10.1136/gut.2008.155812. Epub 2008 Oct 2.
11. Rutgeerts P, Sandborn WJ, Feagan BG et al. Infliximab for induction and maintenance therapy for ulcerative colitis. N Engl J Med 2005; 353 (23): 2462–2476.
12. Rutgeerts P, Feagan BG, Lichtenstein GR et al. Comparison of scheduled and episodic treatment strategies of infliximab in Crohn’s disease. Gastroenterology 2004; 126 (2): 402–413.
13. Present DH, Rutgeerts P, Targan S et al. Infliximab for the treatment of fistulas in patients with Crohn’s disease. N Engl J Med 1999; 340 (18): 1398–1405.
14. Bortlik M, Duricova D, Malickova K et al. Infliximab trough levels may predict sustained response to infliximab in patients with Crohn’s disease. J Crohns Colitis 2013; 7 (9): 736–743. doi: 10.1016/j.crohns.2012.10.019.
15. Billiet T, Cleynen I, Ballet V et al. Prognostic factors for long-term infliximab treatment in Crohn’s disease patients: a 20-year single centre experience. Aliment Pharmacol Ther 2016; 44 (7): 673–683. doi: 10.1111/apt.13754.
16. Papamichael K, Van Stappen T, Vande Casteele N et al. Infliximab Concentration thresholds during induction therapy are associated with short-term mucosal healing in patients with ulcerative colitis. Clin Gastroenterol Hepatol 2016; 14 (4): 543–549. doi: 10.1016/j.cgh.2015.11.014.
17. Ungar B, Levy I, Yavne Y et al. Optimizing anti-TNF-a therapy: serum levels of infliximab and adalimumab are associated with mucosal healing in patients with inflammatory bowel diseases. Clin Gastroenterol Hepatol 2016; 14 (4): 550–557. doi: 10.1016/j.cgh.2015.10.025.
18. Davidov Y, Ungar B, Bar-Yoseph H et al. Association of induction infliximab levels with clinical response in perianal Crohn’s disease. J Crohns Colitis 2016. In press.
19. Yarur AJ, Jain A, Sussman DA et al. The association of tissue anti-TNF drug levels with serological and endoscopic disease activity in inflammatory bowel disease: the ATLAS study. Gut 2016; 65 (2): 249–255. doi: 10.1136/gutjnl-2014-308099.
20. Ben-Horin S, Yavzori M, Benhar I et al. Cross-immunogenicity: antibodies to infliximab in Remicade-treated patients with IBD similarly recognise the biosimilar Remsima. Gut 2016; 65: 1132–1138. doi: 10.1136/gutjnl-2015-309290.
21. Gils A, Van Stappen T, Dreesen E et al. Harmonization of infliximab and anti-infliximab assays facilitates the comparison between originators and biosimilars in clinical samples. Inflamm Bowel Dis 2016; 22: 969–975. doi: 10.1097/MIB. 0000000000000709.
22. Malíčková K, Ďuricová D, Bortlík M et al. Serum trough infliximab levels: a comparison of three different immunoassays for the monitoring of CT-P13 (infliximab) treatment in patients with inflammatory bowel disease. Biologicals 2016; 44 (1): 33–36. doi: 10.1016/j.biologicals.2015.09. 005.
23. Jha A, Upton A, Dunlop WC et al. The budget impact of biosimilar infliximab (Remsima®) for the treatment of autoimmune diseases in five European countries. Adv Ther 2015; 32 (8): 742–756. doi: 10.1007/s12325-015-0233-1.
24. Brodszky V, Rencz F, Péntek M et al. A budget impact model for biosimilar infliximab in Crohn’s disease in Bulgaria, the Czech Republic, Hungary, Poland, Romania, and Slovakia. Expert Rev Pharmacoecon Outcomes Res 2016; 16 (1): 119–125. doi: 10.1586/14737167.2015.1067142.