Abstract
Invasive fungal infections are among the serious infectious complications in liver transplant recipients, with high morbidity and mortality rates. Aspergillosis is the second most common invasive mycosis, and in most cases, occurs in a disseminated form involving multiple organs. Isolated CNS involvement is considered a rare complication. In this case report, we present a patient who underwent liver transplantation and developed headaches in the early post-transplant period, which were later accompanied by visual disturbances. Several months after the onset of symptoms, a definitive gnosis was established only after histological examination of a lesion located on the sphenoid bone of the skull, confirming an infection caused by Aspergillus spp. The patient was started on antifungal therapy according to the infectious disease specialist’s recommendations, with subsequent frequent monitoring of immunosuppressant levels during outpatient follow-up.
This article is an English translation of the original Czech publication. The translation was generated with the assistance of artificial intelligence and has been reviewed and approved by the authors. The original version remains the authoritative source.
Introduction
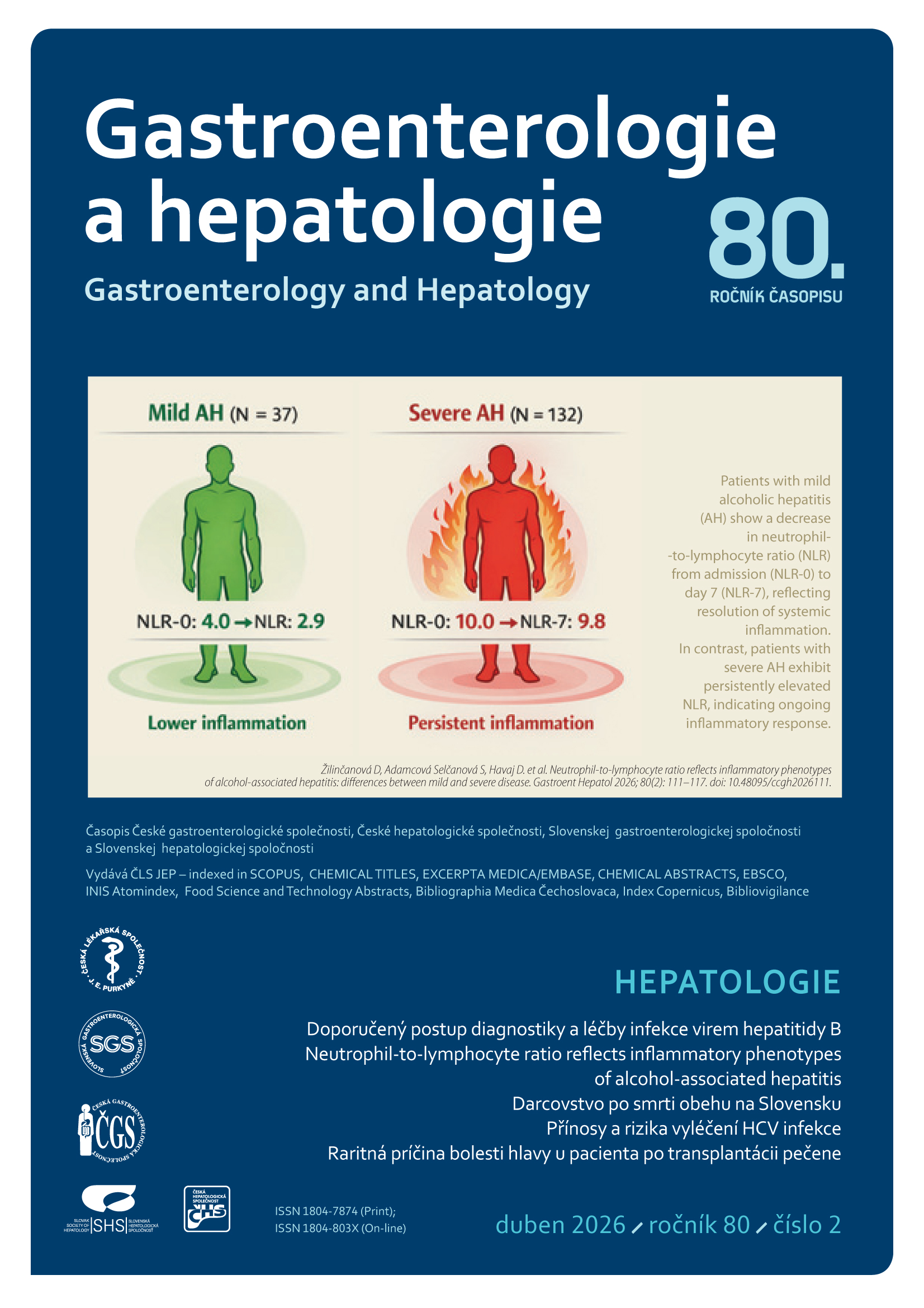
Orthotopic liver transplantation is a life-saving procedure for patients with end-stage chronic liver disease, acute liver failure, and certain types of malignancies [1]. Due to immunosuppressive therapy, post-transplant patients are at risk for several complications, including rejection and infections. Infections are the most common cause of death in the early postoperative period [2]. In addition to common infections, patients are also susceptible to opportunistic infections, including multidrug-resistant microorganisms and invasive fungal infections (IFIs). The incidence of IFI in liver transplant patients ranges from 5% to 42%, with mortality ranging from 25% to 80%. Potential risk factors for the development of IFI can be divided into three groups based on the timing of occurrence: preoperative, intraoperative, and postoperative [3]. A detailed overview of risk factors is provided in Table 1.
Candidiasis is the most common invasive mycosis in patients following solid organ transplantation, particularly of the small intestine, pancreas, and liver, as Candida spp. is a common commensal of the human digestive tract. Among invasive candidiasis cases, the most common pathogen is Candida albicans; however, current epidemiological trends show an increasing prevalence of non-albicans species, such as Candida glabrata, Candida crusei, and Candida auris. Invasive aspergillosis is the second most common invasive mycosis, and its incidence depends on the type of transplanted organ, i.e., lung (8.6%), liver (4.7%), pancreas and kidney (4%), and heart (3.4%). The most frequently isolated species, regardless of the type of transplanted organ, is Aspergillus fumigatus, followed by the less common Aspergillus flavus, Aspergillus niger, and Aspergillus terreus. Cryptococcosis rounds out the top three most frequent IFIs, occurring in approximately 8% of patients following solid organ transplantation. Most cases involve patients after kidney transplantation, less frequently after liver transplantation. The main causative agents are Cryptococcus neoformans and Cryptococcus gattii stricto sensu [4]. Invasive mycoses represent a serious problem in transplant patients and require early diagnosis and treatment.
Case Report
In this case report, we present the case of a 68-year-old male with a history of type 2 diabetes mellitus, who underwent sigmoid resection for adenocarcinoma in 2013 —long-term in remission, with liver cirrhosis due to MetALD at stage CHP C and a MELD score of 17—who underwent a liver transplantation in July 2022 at the Transplant Center of the F. D. Roosevelt University Hospital and Polyclinic in Banská Bystrica. The procedure was uneventful; the patient was gradually started on immunosuppressive therapy, and realimentation and rehabilitation were initiated. During hospitalization, a follow-up abdominal ultrasound revealed a fluid collection in the hepatic hilum, which, following an abdominal CT and MRCP, led to a suspicion of a hepatic biloma. Subsequently, biliary drainage was performed in the angiography suite, and ERCP with placement of a DB drain was performed in the endoscopy suite. Following comprehensive treatment, the patient’s condition improved both clinically and on laboratory tests, and he was discharged to outpatient care with planned removal of the DB drain in 3 months.
In December of the same year, the patient was admitted to the Neurology Department of the J. A. Reiman University Hospital in Prešov due to severe headaches that began in the period following liver transplantation, when the patient was already under outpatient care. Initially, the headaches were diffuse and gradually shifted to the right side of the face. Pain relief was always only temporary following analgesics, requiring repeated adjustments to treatment, eventually necessitating the use of opioids. During hospitalization in the neurology department, the patient underwent several examinations: brain CT angiography without pathology, brain MRI showing isolated demyelinating lesions in the white matter and findings of maxillary and sphenoid sinusitis, carotid ultrasound without hemodynamically significant stenoses, normal EEG, and a diagnostic lumbar puncture with no pathological findings. The patient’s condition was diagnosed as right-sided hemicrania of the cluster headache type. During hospitalization, intravenous analgesic therapy and corticosteroids were initiated, leading to gradual improvement and alleviation of the patient’s symptoms, followed by discharge to outpatient care.
In February 2023, the patient was readmitted, this time to the Department of Ophthalmology at J. A. Reiman University Hospital in Prešov due to sudden onset of amaurosis in the right eye. At the start of hospitalization, a new brain MRI was performed, revealing isolated hyperintense lesions in the white matter of the brain bilaterally, predominantly in the paraventricular and subcortical regions. The condition was assessed as suspected posterior ischemic optic neuropathy. Pulse therapy with corticosteroids and vasodilator therapy were initiated, resulting in partial improvement of vision in the right eye and the patient’s subsequent discharge.
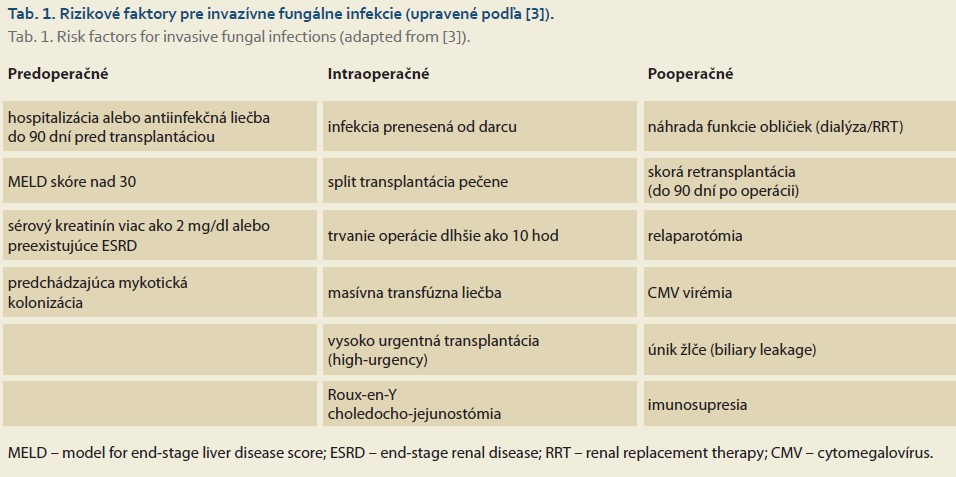
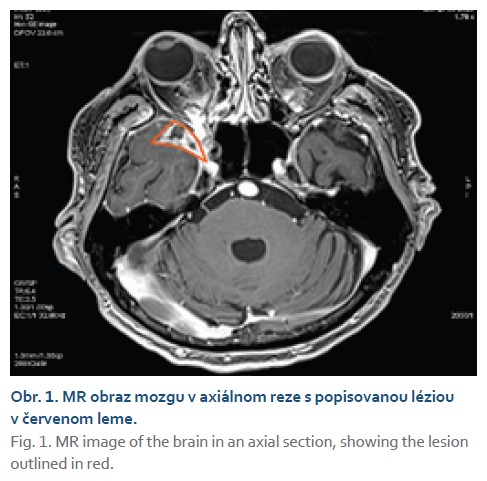
In March 2023, the patient was referred to the Department of Hepatology, Gastroenterology, and Transplantation (HEGITO) at the F. D. Roosevelt University Hospital in Banská Bystrica for further evaluation. The patient continued to experience headaches of a rather episodic nature, particularly at night, and a few days before hospitalization, the patient again experienced loss of vision in the right eye. At the onset of hospitalization, we performed an emergency contrast-enhanced CT scan of the brain, which revealed a enhancing lesion measuring 20 × 13 mm located on the right greater sphenoid wing, extending into the orbit and into the right optic canal, where it partially involved the dorsal quarter of the optic nerve. Subsequently, a follow-up brain MRI confirmed the
aforementioned lesion with involvement of the optic nerve and, to a lesser extent, the medial rectus muscle of the orbit. Based on imaging findings, radiologists leaned toward a diagnosis of a suspected tumor likely meningioma; the possibility of an abscess was less likely (Figs. 1, 2).
In quick succession, we conducted an ophthalmological consultation during hospitalization, which confirmed amaurosis in the right eye and optic disc edema. The consulting neurologist recommended initiating anti-edema and analgesic treatment and arranging a neurosurgical consultation. Given the unruined possibility of infection, we concurrently initiated empirical antibiotic and antifungal therapy (amphotericin B). An ENT consultation ruled out the presence of a focus in the paranasal sinuses. A complete serological workup, including galactomannan testing, was negative in the patient. After consultation with a neurosurgeon, we performed MR spectroscopy, which raised suspicion of an abscess involving the leptomeninges. Given this finding, the patient was referred to the Neurosurgery Department for histological sampling of the described lesion. Upon receiving the histological results, we definitively diagnosed the condition as a fungal brain infection caused by Aspergillus spp. (Fig. 3).
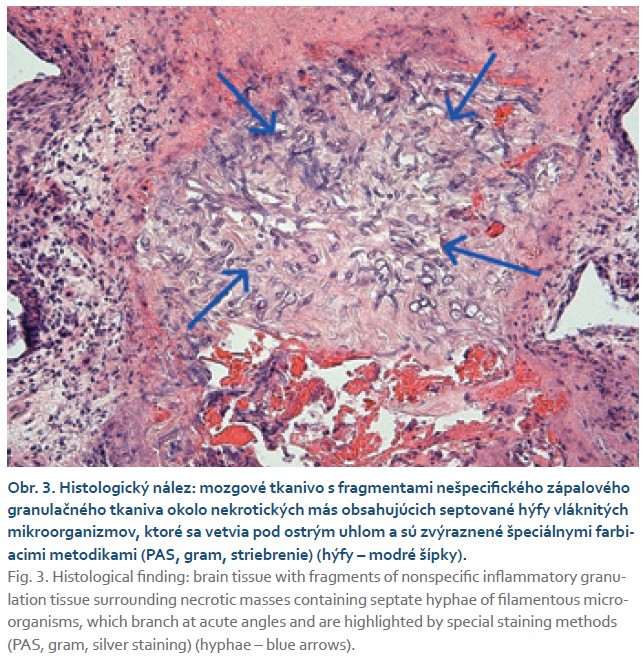
After consultation with an infectious disease specialist, we switched the antifungal treatment to Voriconazole. During hospitalization, the patient showed no laboratory or morphological changes in the graft. We continued immunosuppressive therapy by maintaining Prograf under strict monitoring of its levels; we temporarily discontinued Myfenax, and corticosteroids were gradually tapered off. We discharged the patient to outpatient care with the need for long-term antifungal treatment and regular follow-ups at the neurosurgery outpatient clinic.
Discussion
Invasive aspergillosis (IA) is one of the most common invasive fungal infections in patients after solid organ transplantation (SOT) and is a significant cause of post-transplant morbidity and mortality [5]. The incidence and mortality of IA in liver transplant (LT) patients have decreased over the past decades, from 10% to 1.8% and from 100% to 50%, although mortality following transplantation of other organs remains high. Risk factors that may predispose to IA are consistent with those for invasive mycoses in general [6]. When we look at our patient, we can reliably identify two risk factors, namely from the group of factors associated with postoperative care in the sense of the presence of a hepatic biloma in the early post-transplant period and immunosuppressive therapy.
Invasive aspergillosis occurs in most cases in the early post-transplant period (within 3 months) compared to other organs, with the exception of the heart. At the same time, IA in LT recipients more frequently occurs in a disseminated form (approximately 50% of cases) compared to other types of SOT. Disseminated invasive aspergillosis can affect the lungs, brain, kidneys, liver, or skin (Fig. 4) [6].
Isolated CNS involvement is considered a rare complication, and only a few case reports have been published to date. The cause may be either direct invasion of the CNS or hematogenous spread [7]. The clinical presentation of Aspergillus spp. CNS infection is variable and may include brain abscesses, meningitis, cerebritis, ventriculitis, or thrombosis of the cranial sinuses. Patients may thus exhibit a wide range of symptoms, such as fever, headache, dizziness, lethargy, altered mental status, abnormal gait, or seizures [8]. In our patient, the dominant symptom from the outset was headache, which was later accompanied by severe visual disturbances. It should be noted, however, that headaches are relatively common in post-transplant patients and are usually associated with the use of immunosuppressants, particularly calcineurin inhibitors (tacrolimus, cyclosporine). The incidence of headaches associated with tacrolimus use is reported to be around 32% and is generally associated with high drug levels. However, if headaches persist, further investigation is necessary to rule out other causes, such as newly developed hypertension, infections, or malignancies [9].
The diagnosis of cerebral aspergillosis relies primarily on imaging studies, such as contrast-enhanced CT and MRI of the brain, and histological or culture evidence of Aspergillus spp. in the examined tissue/biological material [8]. Conventional cerebrospinal fluid (CSF) tests are relatively insensitive for the diagnosis of CNS aspergillosis. However, next-generation sequencing (NGS) is emerging, according to several studies, as a promising method for identifying pathogens affecting the CNS, including aspergillosis [10]. Serological testing for galactomannan also yields only moderate results. The sensitivity and specificity for the diagnosis of invasive aspergillosis are 61% and 93%, respectively, according to data from meta-analyses [11]. At the same time, it appears that galactomannan testing is more useful in patients who have undergone hematopoietic stem cell transplantation compared to patients who have undergone solid organ transplantation [12]. In our case as well, the initial cerebrospinal fluid and serum galactomannan testing was inconclusive, and a definitive diagnosis was only established through histological examination of the lesion.
Antifungal treatment with triazoles, particularly voriconazole, is considered the first-line treatment for aspergillosis due to its proven ability, demonstrated in multiple studies, to reduce overall mortality in these patients compared to other antifungals such as amphotericin B [8]. Its dominance is further underscored by excellent penetration into the CNS, efficacy, and good tolerability [6]. However, antifungals carry certain risks, particularly in transplant patients, which must be kept in mind. The most common adverse effects of voriconazole therapy include:
- elevated liver enzymes (in most cases, this involves asymptomatic changes in liver function tests; however, it is essential to monitor liver parameters both before starting voriconazole treatment and then frequently during the first two weeks after treatment initiation, followed by every 2 to 4 weeks);
- visual disturbances (blurred vision, night blindness, decreased visual acuity, etc.);
- skin rashes;
- cardiovascular adverse effects (QT interval prolongation);
- hematological toxicity (anemia, leukopenia/thrombocytopenia) [13].
Antifungals are also characterized by numerous drug interactions, particularly with immunosuppressants. Triazoles act as cytochrome P450 inhibitors, which may increase the levels of co-administered immunosuppressants. It is therefore recommended that the dose of calcineurin inhibitors (tacrolimus) should be reduced by 50–60% of the original dose at the start of treatment with voriconazole, followed by regular monitoring of its levels. The use of sirolimus is contraindicated during treatment with voriconazole.
Mycophenolic acid and corticosteroids do not require dosage adjustment during concomitant treatment with voriconazole [14].
The use of antifungals in the primary prevention of infections caused by invasive mycoses in patients undergoing solid organ transplantation remains a controversial topic. According to current recommendations, antifungal prophylaxis prior to liver transplantation is considered for high-risk patients, specifically those undergoing re-transplantation or repeated abdominal surgery following liver transplantation. However, the optimal duration of antifungal prophylaxis has not been clearly established. This highlights the need for further randomized controlled trials to investigate the impact of antifungal agents on reducing the incidence of invasive mycoses [6].
Conclusion
Invasive aspergillosis is a rare but often fatal complication in patients after liver transplantation. Early diagnosis and immediate treatment are therefore crucial. Voriconazole is the drug of choice, but strict monitoring of immunosuppressant levels is required during its use to avoid overdose. Through our case report, we also aim to highlight the need for early consultation with a transplant center for every patient following a liver transplant, as even a common symptom such as a headache may conceal a rare underlying cause.
ORCID autorov
N. Kubánek 0000-0001-9905-7741
S. Adamcová Selčanová 0000-0001-8181-1937
D. Žilinčanová 0009-0004-7100-0654
K. K. Šulejová 0009-0005-4149-3694
D. J. Havaj 0000-0001-5979-8326
T. Koller 0000-0001-7418-0073
Ľ. Skladaný 0000-0001-5171-3623
Doručené/Submitted: 18. 3. 2026
Prijaté/Accepted: 24. 3. 2026
Korešpondenčná autorka
MUDr. Natália Kubánek
II. Interná klinika a HEGITO
SZU a FNsP F.D.Roosevelta Banská Bystrica
Nám. L. Svobodu 1
975 17 Banská Bystrica
natalia.bystrianska21@gmail.com
Literatúra
1. Adam R, Karam V, Delvart V et al. Evolution of indications and results of liver transplantation in Europe. A report from the European Liver Transplant Registry (ELTR). J Hepatol 2012; 57(3): 675–688. doi: 10.1016/j.jhep.2012.04.015.
2. Agostini C, Buccianti S, Risaliti M et al. Complications in post-liver transplant patients. J Clin Med 2023; 12(19): 6173. doi: 10.3390/jcm12196173.
3. Senoner T, Breitkopf R, Treml B et al. Invasive fungal infections after liver transplantation. J Clin Med 2023; 12(9): 3238. doi: 10.3390/jcm12093238.
4. Mahmoud DE, Hérivaux A, Morio F et al. The epidemiology of invasive fungal infections in transplant recipients. Biomed J 2024; 47(3): 100719. doi: 10.1016/j.bj.2024.100719.
5. Farahani A, Ghiasvand F, Davoudi S et al. Invasive aspergillosis in liver transplant recipients, an infectious complication with low incidence but significant mortality. World J Transplant 2023; 13(5): 264–275. doi: 10.5500/wjt.v13.i5.264.
6. Melenotte C, Aimanianda V, Slavin M et al. Invasive aspergillosis in liver transplant recipients. Transpl Infect Dis 2023; 25(3): e14049. doi: 10.1111/tid.14049.
7. Abbasi M, Ardakani SS, Fakhar N et al. Catastrophic brain aspergillosis after liver transplantation. Case Rep Transplant 2021; 2021: 8626057. doi: 10.1155/2021/8626057.
8. Khodashahi R, Akhavan Rezayat K, Abdollahzade A et al. CNS fungal infections in liver transplantation. Arch Clin Infect Dis 2024; 19(1): e138411. doi: 10.5812/archcid-138411.
9. Montenovo MI, Jalikis FG, Hoch B et al. A symptomatic de novo pheochromocytoma 23 years after liver transplantation: a case report and review of the literature. Case Rep Transplant 2014; 2014: 934385. doi: 10.1155/2014/934385.
10. Chen F, Zhao Y, Shen C et al. Next generation sequencing for gnosis of central nervous system aspergillosis in liver transplant recipients. Ann Transl Med 2021; 9(13): 1071. doi: 10.21037/atm-21-92.
11. Leung V, Stefanovic A, Sheppard D. Severe cerebral aspergillosis after liver transplant. Transpl Infect Dis 2010; 12(1): 51–53. doi: 10.1111/j.1399-3062.2009.00461.x.
12. Pfeiffer CD, Fine JP, Safdar N. Diagnosis of invasive aspergillosis using a galactomannan assay: a meta-analysis. Clin Infect Dis 2006; 42(10): 1417–1427. doi: 10.1086/503427.
13. Eiden C, Peyrière H, Cociglio M et al. Adverse effects of voriconazole: analysis of the French Pharmacovigilance Database. Ann Pharmacother 2007; 41(5): 755–763. doi: 10.1345/aph.1H671.
14. Barchiesi F, Mazzocato S, Mazzanti S et al. Invasive aspergillosis in liver transplant recipients: epidemiology, clinical characteristics, treatment, and outcomes in 116 cases. Liver Transpl 2015; 21(2): 204–212. doi: 10.1002/lt.24032.
15. Gemini. Prompt: Pacient po transplantácii pečene s invazívnou aspergilózou a postihnutím orgánov: pečeň, mozog, obličky, pľúca a koža. 2026 [online]. Dostupné z: https: //gemini.google.com.