Abstract
Background and aims: Alcohol-related liver disease (ALD) represents one of the leading causes of advanced chronic liver disease worldwide and is the predominant etiology of cirrhosis in many European regions. Alcohol-associated hepatitis (AH) represents an inflammatory manifestation of ALD and encompasses a wide spectrum of disease severity ranging from mild disease to severe alcohol-associated hepatitis (SAH), which is characterized by intense systemic inflammation and high short-term mortality. The neutrophil-to--lymphocyte ratio (NLR) has emerged as a simple marker of systemic inflammatory activation and has been associated with adverse outcomes in patients with advanced liver disease. However, its distribution across different clinical phenotypes of AH remains insufficiently characterized. The aim of this study was therefore to evaluate differences in NLR values between mild AH and SAH, and to assess the evolution of NLR during hospitalization. Methods: Using our prospective RH7 registry, we analyzed patients hospitalized with cirrhosis and acute decompensation (AD). Among 1,109 patients enrolled in the registry, 283 fulfilled criteria for AD. The mean age of the cohort was 51.3 ± 12.1 years, and 59% were men. ALD was the most frequent etiology of cirrhosis (207 patients, 73.1%). Acute AH was identified as the precipitating event of decompensation in 169 patients. According to disease severity, 37 patients were classified as having mild AH and 132 fulfilled criteria for SAH. NLR was assessed at admission (NLR-0) and on day 7 of hospitalization (NLR-7). Results: Patients with SAH demonstrated significantly higher inflammatory activity compared to those with mild disease. The mean NLR-0 was 4.0 ± 2.5 in the mild AH group and 10.0 ± 9.6 in the SAH group (P < 0.001). Similar differences were observed on day 7 of hospitalization, when the mean NLR-7 was 2.9 ± 1.8 in mild AH and 9.8 ± 8.6 in SAH (P < 0.001). NLR values remained consistently higher in patients with SAH compared to those with mild AH both at admission and on day 7 of hospitalization. Clinical outcomes differed markedly between the two phenotypes. Thirty-day mortality was 2.7% in mild AH and 30% in SAH. At 90 days, mortality increased to 14% and 46%, respectively, while 1-year mortality reached 24% in mild AH and 55% in SAH. Conclusions: NLR differs substantially between clinical phenotypes of AH and is markedly elevated in SAH. Persistent elevation of NLR during hospitalization likely reflects sustained systemic inflammation characteristic of severe disease. These findings suggest that NLR may serve as a simple and widely available marker of inflammatory activity in patients with AH.
Introduction
Alcohol-related liver disease (ALD) represents one of the most frequent causes of advanced chronic liver disease world--wide and remains the leading etiology of cirrhosis in many European regions [1,2]. The clinical course of ALD is heterogeneous and includes a broad spectrum of manifestations ranging from steatosis and compensated cirrhosis to inflammatory syndromes associated with acute deterioration of liver function [3,4]. Among these manifestations, alcohol-associated hepatitis (AH) represents a distinct clinical entity characterized by acute hepatic inflammation occurring in patients with underlying chronic liver injury [5].
The clinical severity of AH varies considerably. While some patients present with relatively mild disease and favorable clinical course, others develop severe alcohol-associated hepatitis (SAH), a syndrome characterized by profound systemic inflammation, rapid deterioration of liver function, and high short-term mortality [3–5]. The pathophysiology of SAH involves complex interactions between hepatocellular injury, bacterial translocation, endotoxemia, and dysregulated immune activation [6–8]. This inflammatory cascade contributes not only to hepatic injury, but also to systemic complications that may lead to acute decompensation (AD), and in some patients, to acute-on-chronic liver failure (ACLF) [7,8].
Systemic inflammation has emerged as a central mechanism underlying the progression of advanced chronic liver disease and its acute complications [6–8]. Increasing evidence suggests that markers reflecting inflammatory activation may provide important information about disease severity and prognosis in patients with cirrhosis and AD [9–13]. Among these markers, the neutrophil-to-lymphocyte ratio (NLR), calculated from routine differential blood counts, has gained increasing attention as a simple and readily available indicator of systemic inflammatory activity [9].
NLR reflects the balance between activation of the innate immune system, represented by neutrophils, and adaptive immune regulation meted by lymphocytes. Elevated NLR values have been associated with adverse outcomes in a wide range of clinical conditions, including cardiovascular disease, malignancies, sepsis, and chronic inflammatory disorders [10–12,14,15]. In hepatology, NLR has been shown to correlate with disease severity and mortality in patients with cirrhosis, hepatocellular carcinoma, and AD of advanced chronic liver disease [13,16–18].
Despite the increasing interest in NLR as a marker of systemic inflammation in liver disease, limited data exist regarding its distribution across different inflammatory phenotypes within ALD. In particular, it remains unclear whether NLR differs between patients with mild AH and those with SAH, and whether the dynamics of NLR during early hospitalization reflect differences in inflammatory activity between these clinical phenotypes. Therefore, the aim of the present study was to evaluate differences in NLR values between patients with mild AH and SAH and to describe NLR values during the first week of hospitalization.
Methods
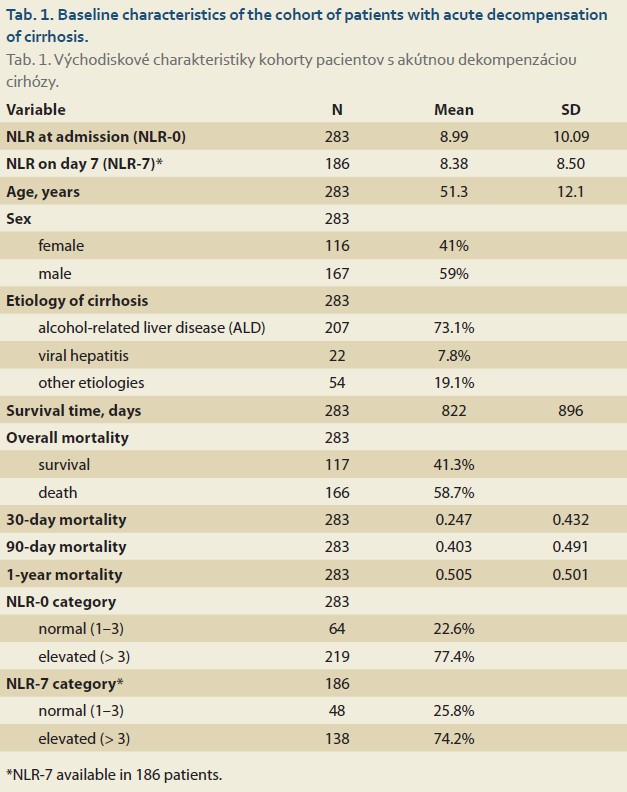
We analyzed patients enrolled in the prospective RH7 registry who were hospitalized for liver cirrhosis and AD. Among 1,109 patients included in the registry, 283 fulfilled criteria for AD and constituted the study cohort (Tab. 1). ALD represented the most frequent etiology of cirrhosis in the cohort. Within this subgroup, AH was identified as the precipitating event of decompensation in 169 patients. According to disease severity, 37 patients were classified as having mild AH and 132 fulfilled criteria for SAH. NLR was assessed at admission (NLR-0) and on day 7 of hospitalization (NLR-7).
For the statistical analysis, we used the R computing environment. Continuous variables were compared using a t-test or Mann-Whitney U test, and categorical variables used the Chi-square or Fisher’s exact test, as deemed appropriate. A P-value < 0.05 was considered statistically significant.
All procedures involving human participants were conducted in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments, as well as the Principles of the Declaration of Istanbul. All patients provided written informed consent prior to enrollment in the RH7 registry. The study protocol was approved by the local ethics committee on May 21, 2014.
Results
Among 1,109 patients with liver cirrhosis enrolled in the RH7 registry, 283 patients (25.5%) fulfilled the criteria for AD and were included in the analysis (Tab. 1). The mean age of the cohort was 51.3 ± 12.1 years (range 22–79 years). Men represented the majority of the population (167 patients, 59%), while 116 patients (41%) were women. With regard to the etiology of underlying cirrhosis, ALD was the most frequent cause and was present in 207 patients (73.1%), followed by other etiologies in 54 patients (19.1%) and viral hepatitis in 22 patients (7.8%). Because ALD represented the predominant etiology in the cohort, further analyses focused on this subgroup.
In the overall AD cohort, the mean NLR-0 at admission was 8.99 ± 10.09, with values ranging from 0.002 to 65.07. Among patients with available follow-up measurements, the mean NLR-7 measured on day 7 of hospitalization was 8.50 ± 8.50, with values ranging from 0.27 to 51.61. Elevated NLR values were common in the study population. Using the predefined threshold, elevated NLR-0 values were observed in 219 patients (77.4%), while 64 patients (22.6%) had NLR values within the normal range. Similarly, among patients with available day-7 measurements, 138 patients (74.2%) had elevated NLR-7 values, whereas 48 patients (25.8%) had values within the normal range.
Within the subgroup of patients with ALD, AH represented the most frequent precipitating event leading to hospitalization for AD. Overall, 169 patients were identified in whom AH was recorded as the trigger of decompensation. According to clinical severity, 37 patients were classified as having mild AH, while 132 patients fulfilled the criteria for SAH (Tab. 2).
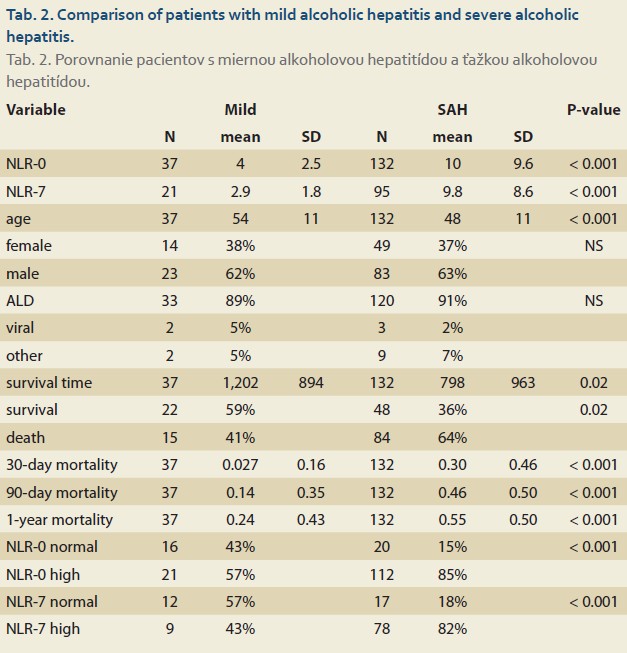
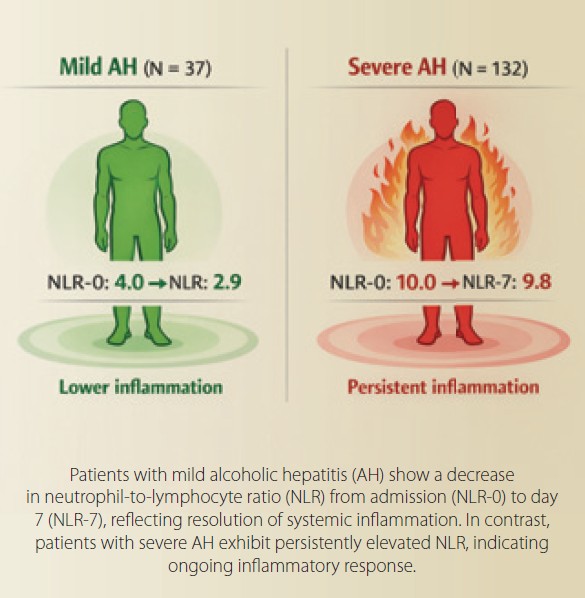
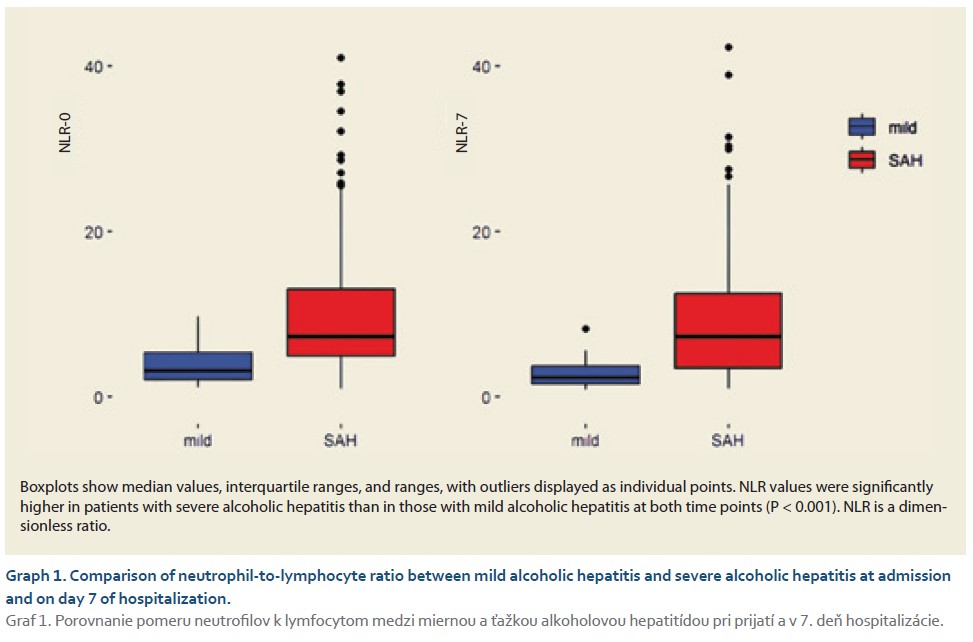
Significant differences in NLR values were observed between these two phenotypes. Patients with mild AH had substantially lower NLR values compared to patients with SAH. The mean NLR-0 was 4.0 ± 2.5 in the mild AH group and 10.0 ± 9.6 in the SAH group (P < 0.001). Similar differences were observed on day 7 of hospitalization, when the mean NLR-7 was 2.9 ± 1.8 in patients with mild AH and 9.8 ± 8.6 in patients with SAH (P < 0.001) (Graph 1). When evaluating the evolution of NLR during hospitalization, a decline in NLR values was observed in patients with mild AH between admission and day 7, suggesting a reduction in systemic inflammatory activity during the early clinical course. In patients with SAH, NLR values remained elevated during hospitalization.
Clinical outcomes differed markedly between the two phenotypes of AH. Patients with SAH had a substantially worse prognosis compared to those with mild disease. Thirty-day mortality was 2.7% in patients with mild AH and 30% in those with SAH. At 90 days, mortality increased to 14% in the mild AH group and 46% in the SAH group, while 1-year mortality reached 24% and 55%, respectively.
Discussion
The present study demonstrates that the NLR differs substantially across clinical phenotypes of AH and is markedly elevated in patients with SAH. In our cohort of patients hospitalized with acute decompensation of cirrhosis, AH represented the most frequent precipitating event, and a clear gradient of systemic inflammatory activity was observed between mild AH and SAH. Patients with SAH had significantly higher NLR values both at admission and on day 7 of hospitalization, whereas patients with mild disease demonstrated considerably lower inflammatory activity and a significant decline in NLR during the early course of hospitalization.
These findings are consistent with the current understanding of the pathophysiology of ALD, in which systemic inflammation represents a central mechanism driving disease progression and clinical deterioration [6–8]. AH, particularly in its severe form, is characterized by intense activation of the innate immune system, neutrophil recruitment into the liver, and increased production of pro-inflammatory cytokines. In this context, the NLR may reflect the imbalance between the pro-inflammatory arm of the immune response, represented by neutrophils, and the adaptive immune compartment represented by lymphocytes [9–11]. Elevated NLR values therefore likely capture the magnitude of systemic inflammatory activation accompanying SAH.
The markedly higher NLR values observed in patients with SAH in our study are biologically plausible. SAH is associated with hepatocellular injury, endotoxemia, and bacterial translocation from the gut, which together stimulate innate immune responses and promote neutrophil activation [6–8]. At the same time, lymphopenia commonly observed in advanced liver disease reflects immune dysregulation and impaired adaptive immune responses. The combination of neutrophilia and relative lymphopenia leads to a substantial increase in the NLR, which may serve as an integrative marker of inflammatory stress.
Changes in NLR during hospitalization were also observed. In patients with mild AH, NLR values tended to decrease during the first week of hospitalization, suggesting a partial reduction in systemic inflammatory activity during the early clinical course. In patients with SAH, NLR values remained elevated during hospitalization.
Persistent systemic inflammation has been increasingly recognized as a key determinant of adverse outcomes in patients with advanced chronic liver disease and acute decompensation [6–8,16,17]. Previous studies have shown that inflammatory markers correlate with disease severity and mortality in cirrhosis and acute-on-chronic liver failure [13,16,17]. Our findings extend these observations by demonstrating that NLR may also reflect differences in inflammatory phenotypes within ALD itself. The elevated NLR values observed in patients with SAH may therefore reflect ongoing inflammatory injury and immune dysregulation.
Consistent with this interpretation, patients with SAH in our cohort also demonstrated markedly worse clinical outcomes. Mortality rates were substantially higher in this group at all evaluated time points, including 30-day, 90-day, and 1-year mortality. This observation is in line with the well-established natural history of SAH, which remains associated with high short-term mortality despite advances in supportive care [3–5]. The association between elevated NLR and poor outcomes further supports the concept that systemic inflammation plays a central role in determining prognosis in these patients [13–15,18].
From a clinical perspective, NLR has several advantages as a biomarker in patients with AH. Unlike many experimental inflammatory markers, NLR can be easily calculated from routinely available laboratory parameters and does not require specialized testing. Its simplicity, low cost, and immete availability make it particularly attractive for clinical practice and for risk stratification in hospitalized patients with advanced liver disease [9–12]. In addition, repeated measurement of NLR during hospitalization may provide insight into the evolution of systemic inflammatory activity and potentially identify patients with persistent inflammatory activation who are at higher risk of adverse outcomes.
Our study has several limitations that should be acknowledged. First, the analysis was performed within a single-center registry cohort, which may limit the generalizability of the findings. Second, the classification of AH severity was based on clinical and laboratory criteria recorded in the registry, and detailed histological confirmation was not available in all cases. Finally, although the study demonstrates a strong association between NLR and disease severity, causal relationships between systemic inflammation and clinical outcomes cannot be definitively established in an observational design. In addition, the relatively small number of patients with mild AH should also be considered when interpreting the results.
Despite these limitations, our study provides important insights into the inflammatory phenotype of AH and highlights the potential role of NLR as a simple marker of systemic inflammatory activation. In conclusion, NLR differs substantially between mild AH and SAH and remains persistently elevated in patients with severe disease. These findings support the concept that NLR reflects the intensity of systemic inflammation in ALD and may represent a useful tool for the assessment of inflammatory activity in patients with AH.
Conclusion
In this cohort of patients with acute decompensation of cirrhosis, the neutrophil-to-lymphocyte ratio markedly differed between mild and severe alcohol-associated hepatitis. Severe disease was associated with substantially higher NLR values, reflecting more pronounced systemic inflammatory activation. NLR may therefore represent a simple and widely available marker of inflammatory activity in patients with alcohol-associated hepatitis.
Abbreviations
ACLF acute-on-chronic liver failure
AD acute decompensation
AH alcohol-associated hepatitis
ALD alcohol-related liver disease
NLR neutrophil-to-lymphocyte ratio
NLR-0 neutrophil-to-lymphocyte ratio at admission
NLR-7 neutrophil-to-lymphocyte ratio on day 7 of hospitalization
RH7 RH7 registry
SAH severe alcohol-associated hepatitis
P. Jarčuška, Ľ. Skladaný: authors share senior authorship.
ORCID of authors
D. Žilinčanová 0009-0004-7100-0654
S. Adamcová Selčanová 0000-0001-8181-1937
D. Havaj 0000-0001-5979-8326
K. Šulejová 0009-0005-4149-3694
N. Kubánek 0000-0001-9905-7741
M. Mihoková 0000-0001-8976-7045
M. Výbošťok 0000-0003-2704-8621
R. Záhorec 0000-0003-2791-6281
P. Jarčuška 0000-0001-8706-1751
Ľ. Skladaný 0000-0001-5171-3623
Submitted/Doručené: 9. 3. 2026
Accepted/Prijaté: 17. 3. 2026
Corresponding author
Svetlana Adamcová Selčanová, MD
2nd Department of Internal Medicine, Div. HEGITO
Faculty of Medicine, Slovak Medical University
Roosevelt University Hospital, Banská Bystrica
L. Svobodu Sq. 1
974 01 Banská Bystrica
sselcanova@gmail.com
References
1. Arroyo V, Jalan R. Acute-on-chronic liver failure: definition, gnosis, and clinical characteristics. Semin Liver Dis 2016; 36(2): 109–116. doi: 10.1055/s-0036-1583202.
2. Moreau R, Jalan R, Gines P et al. Acute-on-chronic liver failure is a distinct syndrome that develops in patients with acute decompensation of cirrhosis. Gastroenterology 2013; 144(7): 1426.e9–1437.e9. doi: 10.1053/j.gastro.2013.02.042.
3. Lucey MR, Mathurin P, Morgan TR. Alcoholic hepatitis. N Engl J Med 2009; 360(26): 2758–2769. doi: 10.1056/NEJMra0805786.
4. Louvet A, Mathurin P. Alcoholic liver disease: mechanisms of injury and targeted treatment. Nat Rev Gastroenterol Hepatol 2015; 12(4): 231–242. doi: 10.1038/nrgastro. 2015.35.
5. Crabb DW, Im GY, Szabo G et al. Diagnosis and treatment of alcohol-associated liver diseases: 2019 practice guidance from the American Association for the Study of Liver Diseases. Hepatology 2020; 71(1): 306–333. doi: 10.1002/hep. 30866.
6. Albillos A, Lario M, Álvarez-Mon M. Cirrhosis-associated immune dysfunction: distinctive features and clinical relevance. J Hepatol 2014; 61(6): 1385–1396. doi: 10.1016/ j.jhep.2014.08.010.
7. Arroyo V, Moreau R, Jalan R et al. Acute-on-chronic liver failure: a new syndrome that will re-classify cirrhosis. J Hepatol 2015; 62(Suppl 1): S131–S143. doi: 10.1016/j.jhep.2014.11.045.
8. Trebicka J, Fernandez J, Papp M et al. The PREDICT study uncovers three clinical courses of acutely decompensated cirrhosis that have distinct pathophysiology. J Hepatol 2020; 73(4): 842–854. doi: 10.1016/j.jhep.2020.06.013.
9. Zahorec R. Ratio of neutrophil to lymphocyte counts-rapid and simple parameter of systemic inflammation and stress in critically ill. Bratisl Lek Listy 2001; 102(1): 5–14.
10. Forget P, Khalifa C, Defour JP et al. What is the normal value of the neutrophil-to-lymphocyte ratio? BMC Res Notes 2017; 10(1): 12. doi: 10.1186/s13104-016-2335-5.
11. Guthrie GJ, Charles KA, Roxburgh CS et al. The systemic inflammation-based neutrophil-lymphocyte ratio: experience in patients with cancer. Crit Rev Oncol Hematol 2013; 88(1): 218–230. doi: 10.1016/j.critrevonc.2013.03.010.
12. Templeton AJ, McNamara MG, Šeruga B et al. Prognostic role of neutrophil-to-lymphocyte ratio in solid tumors: a systematic review and meta-analysis. J Natl Cancer Inst 2014; 106(6): dju124. doi: 10.1093/jnci/dju124.
13. Biyik M, Ucar R, Solak Y et al. Blood neutrophil-to-lymphocyte ratio independently predicts survival in patients with liver cirrhosis. Eur J Gastroenterol Hepatol 2013; 25(4): 435–441. doi: 10.1097/MEG.0b013e32835c2af3.
14. Chiriac S, Stanciu C, Singeap AM et al. Prognostic value of neutrophil-to-lymphocyte ratio in cirrhotic patients with acute-on-chronic liver failure. Turk J Gastroenterol 2020; 31(12): 868–876. doi: 10.5152/tjg.2020.19838.
15. Moreau R, Clària J, Aguilar F et al. Blood metabolomics uncovers inflammation-associated mitochondrial dysfunction as a potential mechanism underlying ACLF. J Hepatol 2020; 72(4): 688–701. doi: 10.1016/j.jhep.2019.11.009.
16. Laleman W, Claria J, Van der Merwe S et al. Systemic inflammation and acute-on-chronic liver failure: too much, not enough. Can J Gastroenterol Hepatol 2018; 2018: 1027152. doi: 10.1155/2018/1027152.
17. Žilinčanová D, Skladaný Ľ, Jarčuška P et al. Neutrophil-to-lymphocyte ratio in acute decompensation of advanced chronic liver disease and ACLF: prognostic significance by etiology. Clin Exp Hepatol.
18. Šafčák D, Dražilová S, Gazda J et al. Prognostic value of neutrophil-to-lymphocyte ratio across Barcelona clinic liver cancer stages in hepatocellular carcinoma: a multicenter retrospective analysis. Bratisl Med J 2026. doi: 10.1007/s44411-026-00530-4.