Abstract
Chronic hepatitis C virus (HCV) infection remains a major cause of liver-related morbidity and mortality worldwide. The natural course of HCV infection is highly variable, ranging from asymptomatic disease to significant fibrosis, cirrhosis, and hepatocellular carcinoma (HCC). The advent of direct-acting antivirals (DAAs) has revolutionized HCV treatment, achieving sustained virologic response (SVR) rates exceeding 95% across all stages of liver disease. Viral eradication is associated with reduced liver inflammation, regression of fibrosis, and reduced portal hypertension. More importantly, SVR significantly reduces the risk of liver decompensation, HCC, and liver disease-related mortality. However, the risk of HCC is not entirely eliminated, particularly in patients with advanced fibrosis or cirrhosis, and therefore, HCC surveillance is recommended. DAA therapy has also made it possible to treat patients with decompensated cirrhosis, leading to improved liver function, and in selected cases, removal of the patient from the liver transplant waiting list. However, treatment response and clinical course depend on baseline liver function; in patients with advanced disease (MELD score ≥ 20), the chance of achieving SVR is lower, with a higher risk of adverse events. Even after achieving SVR, metabolic risk factors, including obesity, betes, and fatty liver disease, may negatively impact further course of the disease and the risk of developing HCC. Weight gain after achieving SVR can be significant and influenced by host genetic factors, such as variants of the PNPLA3 gene.
This article is an English translation of the original Czech publication. The translation was generated with the assistance of artificial intelligence and has been reviewed and approved by the authors. The original version remains the authoritative source.
Introduction
Chronic hepatitis C (HCV) is one of the leading causes of chronic liver disease in developed countries. According to the latest global data, the prevalence of HCV RNA positivity is approximately 0.7%, representing about 56.8 million infected individuals worldwide [1]. HCV infection remains one of the diseases that increase liver morbidity and mortality [2].
HCV infection can have a highly variable clinical course, ranging from an asymptomatic form to chronic hepatitis with fibrosis, which can lead to liver cirrhosis and its complications, including chronic liver failure and hepatocellular carcinoma (HCC) [3].
The primary goal of treatment is the eradication of the virus from the body. Successful elimination of the virus prevents liver damage, slows or completely halts the progression of fibrosis, and prevents the development of cirrhosis and late complications of end-stage liver disease (decompensated cirrhosis and the development of HCC). Sustained viral eradication, referred to as sustained virologic response (SVR12 or SVR24), means the absence of HCV RNA in the blood 12 or 24 weeks after the end of treatment [4]. Achieving SVR corresponds to a cure of HCV infection, as late relapse occurs in less than 0.2% of cases after 6 months of follow-up [5]. Sustained viral eradication is associated with the regression of inflammatory changes and improvement in liver fibrosis [4,6]. Despite the availability of direct-acting antivirals (DAAs), which are highly effective (over 95%) regardless of the stage of liver disease, it is estimated that only 13–21% of diagnosed patients will initiate DAA treatment [4].
Epidemiology
Hepatitis C (HCV) is a globally widespread infection that, with a prevalence of approximately 0.7% in the general population, remains one of the leading causes of liver disease in developed countries [4]. According to a serological survey from 2001, a prevalence of 0.2% was found in the Czech Republic [7]. More recent data from a 2015 seroprevalence study show a 1.67% prevalence of anti-HCV antibodies, with 0.93% of individuals also testing positive for HCV RNA, indicating active viral replication [8]. Until 2007, genotype 1b was dominant in the Czech Republic [9], while the current distribution of genotypes is as follows: 25% genotype 1a, 25% genotype 1b, and 46.4% genotype 3 [8].
HCV Transmission The primary mode of HCV transmission is contact with infected blood, followed by mother-to-child transmission and sexual transmission. Historically, the most significant route was blood transfusion and blood products (especially before 1992); today, intravenous drug use and sharing of injection equipment are the primary causes [10,11].
Course of the disease
HCV infection is rarely diagnosed during the acute phase. Clinical symptoms usually appear within 7–8 weeks after exposure to HCV RNA; in most patients, the acute phase is completely asymptomatic. Available data indicate that 50–85% of infected individuals are unable to spontaneously clear the virus during the acute phase. The disease then progresses to a chronic stage, arbitrarily defined as an infection lasting longer than 6 months [4,12].
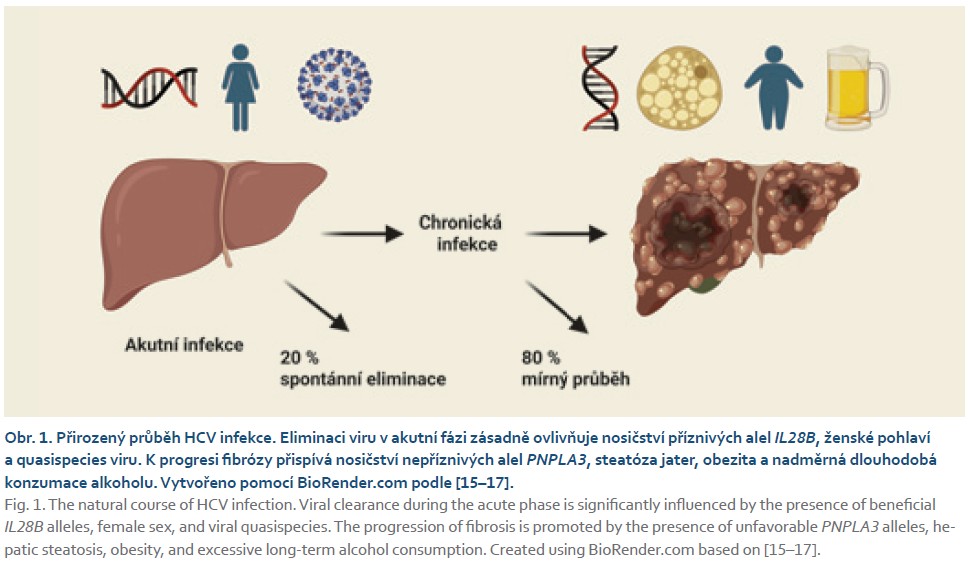
The course of HCV infection is generally mild for years, and its progression over time is slow [12]. Nevertheless, epidemiological studies indicate that the infection is associated with significant mortality [3]. In 15–30% of patients, liver cirrhosis develops within 10–20 years of infection; furthermore, after 20–30 years, 1.8–8.3% of patients develop HCC [13]. In immunocompetent individuals, the disease is usually minimally active for approximately two decades. The highest incidence of cirrhosis is observed after approximately 25 years of infection, at which point approximately 35% of infected individuals develop cirrhosis. Conversely, in patients who maintain normal liver enzyme levels over the long term during infection, the occurrence of cirrhosis has not been reported [3,13]. Conversely, the progression of fibrosis is adversely affected by long-term alcohol abuse, hepatic steatosis, obesity, and carriage of deleterious variants of the PNPLA3 gene (Fig. 1) [14–16].
Benefits of HCV cure and residual risk of disease after achieving SVR
The primary goal of HCV treatment is to prevent the progression of liver fibrosis and, in particular, late complications of the disease, such as liver cirrhosis and HCC. Another goal is to prevent transmission to other healthy individuals, particularly in groups with high infection prevalence, such as people who inject drugs (PWID), men who have sex with men, incarcerated individuals, hospitalized patients, or healthcare workers performing invasive procedures [4].
A cost-effectiveness analysis comparing a “treat-all” strategy with a strategy based on fibrosis stage demonstrated that treatment based on fibrosis stage is not cost-effective; DAA treatment should be provided to all patients regardless of disease stage [18].
Reduction in fibrosis stage and portal hypertension stage
Viral eradication reduces the risk of cirrhosis decompensation and death from liver disease. Studies from the era of IFN treatment demonstrate that even cirrhosis can regress in a significant proportion of patients upon achieving SVR. In 2012, D’Ambrosio et al. demonstrated regression of F4 fibrosis in liver biopsy to lower fibrosis grades following successful antiviral treatment in more than 60% of cases, with a median follow-up of 5 years [19]. Fibrosis regression was also demonstrated in the case of non-invasive fibrosis assessment during DAA treatment: a study by Bachofner et al. demonstrated an early decrease in liver stiffness measured using vibration-controlled transient elastography, while a decrease in serum fibrosis markers (APRI and FIB-4 scores) was also evident [20]. A Spanish study conducted on a cohort of more than 500 patients treated with DAAs confirmed a rapid decrease in liver stiffness following successful treatment: after one year, there was a 30% decrease in liver stiffness [21].
Successful antiviral treatment also affects the degree of portal hypertension. The first study analyzing HVPG (hepatic venous pressure gradient) following DAA treatment was published by Mandorfer et al. in 2016: achieving SVR improves HVPG; however, in patients with HVPG ≥ 16 mmHg, the decrease is less pronounced [22]. The same group of authors later investigated HVPG after achieving SVR in patients with clinically significant portal hypertension prior to treatment. A ≥ 10% reduction in HVPG after treatment was associated with a significantly lower risk of decompensation of liver cirrhosis [23].
In patients with HCV and cirrhosis, the risk of disease decompensation is proportional to the degree of portal hypertension. A reduction in the risk of decompensation was clearly demonstrated after achieving SVR in studies with IFN-based regimens in patients followed for more than 5 years [24]. DAA therapy currently allows for the treatment of patients who could not have been treated in the past, primarily due to anticipated hematologic adverse effects, most commonly thrombocytopenia. Given the relatively short time since the introduction of DAAs into clinical practice, there are only a few significant studies demonstrating treatment outcomes in patients with cirrhosis. A French study did not confirm a lower risk of decompensation in patients treated with DAAs and followed up long-term with a median of 33.4 months [25]. In contrast, Mendizabal’s study, involving a total of 1,760 patients with cirrhosis, demonstrated a lower risk of decompensation following DAA treatment during a 26.2-month follow-up period [26].
Reduction in the risk of HCC and mortality
Active viral replication represents the most significant risk factor for the development of HCC.
Therefore, achieving SVR most effectively reduces the risk of HCC, regardless of whether SVR is achieved through treatment with IFN, IFN + DAA, or DAA alone. Achieving SVR reduces the risk of HCC by 71%, as demonstrated in a study involving 62,000 individuals [27]. The results of the previously described study were also confirmed in patients treated exclusively with DAA. None of the studies confirmed a complete minimization of the risk of HCC; the incidence of HCC remains stable between 1.5 and 2.3 cases per 100 patient-years in individuals with cirrhosis and does not decrease even 3.6 years after achieving SVR [28].
The risk of developing HCC even after successful treatment is due to several mechanisms: the regression of advanced fibrosis and cirrhosis is a long-term process during which the risk of HCC
persists. In some patients with cirrhosis, the remodeling process is no longer possible; in others, fibrosis may progress for other reasons (excessive alcohol consumption, coexisting non-alcoholic steatohepatitis). Another factor is preneoplastic genetic and epigenetic changes that arise even before SVR is achieved and persist for life [29].
Because eradication of HCV infection reduces but does not completely eliminate the risk of HCC, the risk in patients with severe fibrosis (F3 according to the METAVIR score) and cirrhosis (F4) is so high that the recommendation to continue HCC surveillance even after achieving SVR is unequivocal [4,30]. Nevertheless, antiviral treatment of HCV can currently be considered one of the most effective forms of chemoprevention for this malignant disease.
An important benefit of successful treatment is reduced mortality, which is evident in patients with cirrhosis treated with DAAs. In a French cohort of 3,045 patients with cirrhosis, a decrease in all-cause mortality and mortality due to liver disease was observed [25].
Improvement in synthetic liver function: “recompensation” and the possibility of removal from the liver transplant waiting list
Achieving SVR with interferon regimens was nearly impossible in patients with decompensated liver cirrhosis; now it is possible to treat even these patients with DAAs. Treatment may lead to improved liver function. In 50% of patients, improvement is observed with treatment using the combination of sofosbuvir and velpatasvir when assessing the MELD score (Model for End-Stage Liver Disease) 12 weeks after treatment compared to the MELD score prior to initiating therapy [31]. Foster et al. reached the same conclusions in a retrospective study evaluating 409 patients: DAA treatment led to a reduction in the MELD score over the course of 6 months. Patients who had serum albumin concentrations < 35 g/L, Na < 135 mmol/L, and were older than 65 years prior to treatment had a lower chance of improvement [32].
For patients with decompensated liver cirrhosis, DAAs represent an ideal treatment: the treatment is short, has no side effects, and the likelihood of premature discontinuation of therapy is therefore very low [33]. Conversely, the chance of achieving SVR is high, though still lower than in patients with compensated disease, and the risk of adverse effects attributable to treatment increases with the severity of liver disease [34].
In patients treated with a combination of sofosbuvir and ledipasvir in real-world clinical practice, SVR rates depended on the presence of cirrhosis (94%), a history of decompensation (90%), and the use of proton pump inhibitors (94%) [34].
Treatment in a patient with decompensated liver cirrhosis prior to liver transplantation (LTx) aims to improve liver function and prevent HCV recurrence in the liver graft [35]. In patients on the waiting list, improved liver function may lead to removal from the waiting list; viral eradication results in a significant improvement in bilirubin, albumin, and prothrombin time levels in up to one-third of patients [36]. Patients with a Child-Pugh B functional classification benefit more from infection clearance in terms of event-free survival 15 months after treatment compared to cirrhotic patients in the Child-Pugh C functional classification group [32].
Removing a patient from the waiting list upon improvement in liver function is particularly crucial in Western countries, given the shortage of organs suitable for transplantation. In patients removed from the waiting list due to improved liver function, we must continue to monitor liver function and perform HCC screening even after successful treatment [4,30].
Achieving SVR in patients on the LTx waiting list prevents HCV recurrence in the liver graft [37]. Currently, any combination of DAAs may be used, provided that liver function is taken into account. A treatment regimen containing a protease inhibitor is contraindicated in patients with Child-Pugh functional class B and C. The risk of adverse effects stems from high drug concentrations in the blood in the presence of liver dysfunction [4]. Treatment efficacy in patients with decompensated cirrhosis is lower compared to those with compensated cirrhosis. In patients with a MELD score ≥ 15, there is a significantly higher risk of decompensation as well as an increased risk of treatment-related adverse effects [32]. Patients with a MELD score < 16 points prior to treatment who achieve a decrease in MELD of more than 4 points at the end of therapy have the highest chance of improved liver function and removal from the waiting list following successful antiviral treatment. Conversely, in patients with a MELD score ≥ 20, the chance of improved liver function is very low [33]. Patients with a MELD score ≥ 20 should therefore be treated only after LTx, as should individuals with refractory complications of portal hypertension (ascites, encephalopathy) [4].
Weight gain after successful treatment
Patients who have been successfully cured may still have elevated liver enzyme levels even after achieving SVR. Among 834 patients who achieved SVR, 10% had persistently elevated ALT levels [38]; higher ALT levels correlated with higher BMI. Higher ALT levels were also observed in individuals with beta-lactam use and excessive alcohol consumption [39]. Patients with a higher BMI have a higher risk of liver cirrhosis after achieving SVR [40].
Recent studies document weight gain after achieving SVR. In a German study involving 284 patients treated with DAAs, 44% of patients gained weight, averaging 1.45 kg per year after treatment completion. The factor that predicted weight gain was age under 60 years, but not gender, a history of beta-hemoglobinopathy, cirrhosis, or pre-treatment BMI [41]. These results were confirmed by other studies: results from a prospective study of 11,469 U.S. veterans show weight gain in 52.6% of patients: 19.8% of them gained significant weight, defined as an increase of ≥ 9 kg 2 years after treatment [42]. In this study, independent predictors of weight gain included achieving SVR, younger age (< 65 years), cirrhosis, a FIB-4 score > 3.25, and being overweight prior to treatment. Conversely, abstinence from alcohol was a protective factor. The authors conclude from the study that the most likely mechanisms causing weight gain are neuropsychiatric changes and a reduction in the concentration of circulating pro-inflammatory cytokines. Chronic liver disease is often accompanied by loss of appetite and changes in taste; therefore, successful treatment may lead to the resolution of these symptoms and subsequent weight gain.
A study conducted on 230 patients who achieved SVR and were followed for an additional 3 years after HCV treatment with DAAs also recorded an increase in BMI, specifically by 0.39 kg/m2 after 3 years. Weight gain was influenced by the PNPLA3 genotype and was highest in the group carrying at least one PNPLA3 G variant allele; weight gain in this group was 0.94 kg/m2. Carriage of the PNPLA3 G allele was the only factor predicting weight gain after treatment in the multivariate analysis [43].
MASLD as Another Liver Disease
Metabolic dysfunction-associated steatohepatitis (MASLD) is currently the most common liver disease and a significant cause of morbidity and mortality associated with liver diseases [44]; therefore, it often coexists with HCV. Patients with HCV and steatosis are at risk of faster fibrosis
progression and HCC [45]. Achieving viral eradication may lead to the resolution of infection-induced steatosis, particularly in patients with genotype 3; however, a significant proportion of patients have steatosis unrelated to HCV, which can thus contribute to fibrosis progression.
Diabetes mellitus is a major risk factor for the progression of fibrosis in hepatitis C. HCV infection interferes with insulin metabolism, resulting in insulin resistance and the development of diabetes [45]. Successful antiviral treatment can prevent the development of insulin resistance or diabetes, but those who already have diabetes prior to treatment initiation will not achieve normalization of glucose metabolism after achieving SVR; however, they may require lower doses of antiretrovirals [46]. A large-scale study followed a total of 1,000 patients with severe fibrosis and cirrhosis who achieved SVR; the median follow-up was 5.7 years. A total of 51 patients developed HCC, and progression of liver disease was observed in 101 patients. The presence of genotype 2, along with older age and thrombocytopenia, predicted the occurrence of HCC in a multivariate analysis [3].
Conclusion
Thanks to the high efficacy of DAAs, all patients who are aware of their infection and have access to treatment can currently be treated. Elimination of HCV infection, and thus a reduction in the risk of late complications, can be achieved in countries with effective screening programs that target, in particular, groups of people with a high prevalence of the disease. Even individuals who have been successfully cured should remain in the HCC surveillance program if they had severe liver fibrosis or cirrhosis prior to treatment.
ORCID autorky
S. Fraňková 0000-0002-1462-5920
Doručeno/Submitted: 22. 3. 2026
Přijato/Accepted: 27. 3. 2026
Korespondenční autor
MUDr. Tomáš Nesnídal
Klinika hepatogastroenterologie
Institut klinické a experimentální medicíny
Vídeňská 1958/9
140 21 Praha 4
tomas.nesnidal@ikem.cz
Literatura
1. Polaris Observatory HCV collaborators. Global change in hepatitis C virus prevalence and cascade of care between 2015 and 2020: a modelling study. Lancet Gastroenterol Hepatol 2022; 7(5): 396–415. doi: 10.1016/S2468-1253(21)00472-6.
2. Hovaguimian F, Beeler PE, Mullhaupt B et al. Mortality and morbidity related to hepatitis C virus infection in hospitalized adults-A propensity score matched analysis. J Viral Hepat 2023; 30(9): 765–774. doi: 10.1111/jvh.13861.
3. van der Meer AJ, Veldt BJ, Feld JJ et al. Association between sustained virological response and all-cause mortality among patients with chronic hepatitis C and advanced hepatic fibrosis. JAMA 2012; 308(24): 2584–2593. doi: 10.1001/jama.2012.144878.
4. European Association for the Study of the Liver. EASL recommendations on treatment of hepatitis C: final update of the series. J Hepatol 2020; 73(5): 1170–1218. doi: 10.1016/j.jhep.2020.08.018.
5. Sarrazin C, Isakov V, Svarovskaia ES et al. Late relapse versus hepatitis C virus reinfection in patients with sustained virologic response after sofosbuvir-based therapies. Clin Infect Dis 2017; 64(1): 44–52. doi: 10.1093/cid/ciw676.
6. Calvaruso V, Craxi A. Hepatic benefits of HCV cure. J Hepatol 2020; 73(6): 1548–1556. doi: 10.1016/j.jhep.2020.08.006.
7. Nemecek V, Castkova J, Fritz P et al. The 2001 serological survey in the Czech Republic – viral hepatitis. Cent Eur J Public Health 2003; (Suppl 11): S54–S61.
8. Chlibek R, Smetana J, Sosovickova R et al. Prevalence of hepatitis C virus in adult population in the Czech Republic – time for birth cohort screening. PloS One 2017; 12(4): e0175525. doi: 10.1371/journal.pone.0175525.
9. Cornberg M, Razavi HA, Alberti A et al. A systematic review of hepatitis C virus epidemiology in Europe, Canada and Israel. Liver Int 2011; 31(Suppl 2): 30–60. doi: 10.1111/j.1478-3231.2011.02539.x.
10. Frankova S, Jandova Z, Jinochova G et al. Therapy of chronic hepatitis C in people who inject drugs: focus on adherence. Harm Reduct J 2021; 18(1): 69. doi: 10.1186/s12954-021-00519-y.
11. Edlin BR. Perspective: test and treat this silent killer. Nature 2011; 474(7350): S18–S19. doi: 10.1038/474S18a.
12. Kenny-Walsh E. Clinical outcomes after hepatitis C infection from contaminated anti-D immune globulin. Irish Hepatology Research Group. N Engl J Med 1999; 340(16): 1228–1233. doi: 10.1056/NEJM199904223401602.
13. Toshikuni N, Arisawa T, Tsutsumi M. Hepatitis C-related liver cirrhosis – strategies for the prevention of hepatic decompensation, hepatocarcinogenesis, and mortality. World J Gastroenterol 2014; 20(11): 2876–2887. doi: 10.3748/wjg.v20.i11.2876.
14. Llamosas-Falcon L, Shield KD, Gelovany M et al. Impact of alcohol on the progression of HCV-related liver disease: a systematic review and meta-analysis. J Hepatol 2021; 75(3): 536–546. doi: 10.1016/j.jhep.2021.04.018.
15. Frankova S, Uzlova N, Merta D et al. Predictors of significant liver fibrosis in people with chronic hepatitis C who inject drugs in the Czech Republic. Life (Basel) 2023; 13(4): 932. doi: 10.3390/life13040932.
16. Senkerikova R, Frankova S, Jirsa M et al. PNPLA3 rs738409 G allele carriers with genotype 1b HCV cirrhosis have lower viral load but develop liver failure at younger age. PloS One 2019; 14(9): e0222609. doi: 10.1371/journal.pone.0222609.
17. Asselah T, Bieche I, Sabbagh A et al. Gene expression and hepatitis C virus infection. Gut 2009; 58(6): 846–858. doi: 10.1136/gut.2008.166348.
18. Younossi ZM, Singer ME, Mir HM et al. Impact of interferon free regimens on clinical and cost outcomes for chronic hepatitis C genotype 1 patients. J Hepatol 2014; 60(3): 530–537. doi: 10.1016/j.jhep.2013.11.009.
19. D’Ambrosio R, Aghemo A, Rumi MG et al. A morphometric and immunohistochemical study to assess the benefit of a sustained virological response in hepatitis C virus patients with cirrhosis. Hepatology 2012; 56(2): 532–543. doi: 10.1002/hep.25606.
20. Bachofner JA, Valli PV, Kroger A et al. Direct antiviral agent treatment of chronic hepatitis C results in rapid regression of transient elastography and fibrosis markers fibrosis-4 score and aspartate aminotransferase-platelet ratio index. Liver Int 2017; 37(3): 369–376. doi: 10.1111/liv.13256.
21. Pons M, Rodriguez-Tajes S, Esteban JI et al. Non-invasive prediction of liver-related events in patients with HCV-associated compensated advanced chronic liver disease after oral antivirals. J Hepatol 2020; 72(3): 472–480. doi: 10.1016/j.jhep.2019.10.005.
22. Mandorfer M, Kozbial K, Schwabl P et al. Sustained virologic response to interferon-free therapies ameliorates HCV-induced portal hypertension. J Hepatol 2016; 65(4): 692–699. doi: 10.1016/j.jhep.2016.05.027.
23. Mandorfer M, Kozbial K, Schwabl P et al. Changes in hepatic venous pressure gradient predict hepatic decompensation in patients who achieved sustained virologic response to interferon-free therapy. Hepatology 2020; 71(3): 1023–1036. doi: 10.1002/hep.30885.
24. Nahon P, Bourcier V, Layese R et al. Eradication of hepatitis C virus infection in patients with cirrhosis reduces risk of liver and non-liver complications. Gastroenterology 2017; 152(1): 142.e2–156.e2. doi: 10.1053/j.gastro.2016.09.009.
25. Carrat F, Fontaine H, Dorival C et al. Clinical outcomes in patients with chronic hepatitis C after direct-acting antiviral treatment: a prospective cohort study. Lancet 2019; 393(10179): 1453–1464. doi: 10.1016/S0140-6736(18)32111-1.
26. Mendizabal M, Pinero F, Ridruejo E et al. Disease progression in patients with hepatitis C virus infection treated with direct-acting antiviral agents. Clin Gastroenterol Hepatol 2020; 18(11): 2554–2563.e3. doi: 10.1016/ j.cgh.2020.02.044.
27. Ioannou GN, Green PK, Berry K. HCV eradication induced by direct-acting antiviral agents reduces the risk of hepatocellular carcinoma. J Hepatol 2017; S0168-8278(17)32273-0. doi: 10.1016/j.jhep.2017.08.030.
28. Kanwal F, Kramer JR, Asch SM et al. Risk of hepatocellular carcinoma in HCV patients treated with direct acting antiviral agents. Hepatology 2020; 71(1): 44–55. doi: 10.1002/hep.30823.
29. Paradis V, Dargere D, Bonvoust F et al. Clonal analysis of micronodules in virus C-induced liver cirrhosis using laser capture microdissection (LCM) and HUMARA assay. Lab Invest 2000; 80(10): 1553–1559. doi: 10.1038/labinvest. 3780165.
30. European Association for the Study of the Liver. EASL Clinical Practice Guidelines on the management of hepatocellular carcinoma. J Hepatol 2025; 82(2): 315–374. doi: 10.1016/j.jhep.2024.08.028.
31. Curry MP, O’Leary JG, Bzowej N et al. Sofosbuvir and velpatasvir for HCV in patients with decompensated cirrhosis. N Engl J Med 2015; 373(27): 2618–2628. doi: 10.1056/NEJMoa1512614.
32. Foster GR, Irving WL, Cheung MC et al. Impact of direct acting antiviral therapy in patients with chronic hepatitis C and decompensated cirrhosis. J Hepatol 2016; 64(6): 1224–1231. doi: 10.1016/j.jhep.2016.01.029.
33. Belli LS, Berenguer M, Cortesi PA et al. Delisting of liver transplant candidates with chronic hepatitis C after viral eradication: a European study. J Hepatol 2016; 64(6): 1224–1231. doi: 10.1016/j.jhep.2016.05.010.
34. Terrault NA, Zeuzem S, Di Bisceglie AM et al. Effectiveness of ledipasvir-sofosbuvir combination in patients with hepatitis C virus infection and factors associated with sustained virologic response. Gastroenterology 2016; 151(6): 1131–1140 e5. doi: 10.1053/j.gastro.2016.08.004.
35. Fraňková S, Pítová V, Šperl J. Transplantace jater pro chronickou hepatitidu C – význam protivirové léčby v roce 2024. Gastroent Hepatol 2024; 78(2): 108–114. doi: 10.48095/ccgh2024108.
36. Belli LS, Duvoux C, Berenguer M et al. ELITA consensus statements on the use of DAAs in liver transplant candidates and recipients. J Hepatol 2017; 67(3): 585–602. doi: 10.1016/ j.jhep.2017.03.006.
37. Curry MP, Forns X, Chung RT et al. Sofosbuvir and ribavirin prevent recurrence of HCV infection after liver transplantation: an open-label study. Gastroenterology 2015; 148(1): 100–107.e1. doi: 10.1053/j.gastro.2014.09.023.
38. Welsch C, Efinger M, von Wagner M et al. Ongoing liver inflammation in patients with chronic hepatitis C and sustained virological response. PloS One 2017; 12(2): e0171755. doi: 10.1371/journal.pone.0171755.
39. Tacke F, Boeker KHW, Klinker H et al. Baseline risk factors determine lack of biochemical response after SVR in chronic hepatitis C patients treated with DAAs. Liver Int 2020; 40(3): 539–548. doi: 10.1111/liv.14186.
40. Benhammou JN, Moon AM, Pisegna JR et al. Nonalcoholic fatty liver disease risk factors affect liver-related outcomes after direct-acting antiviral treatment for hepatitis C. Dig Dis Sci 2021; 66(7): 2394–2406. doi: 10.1007/s10620-020-06457-2.
41. Schlevogt B, Deterding K, Port K et al. Interferon-free cure of chronic hepatitis C is associated with weight gain during long-term follow-up. Z Gastroenterol 2017; 55(9): 848–856. doi: 10.1055/s-0043-112656.
42. Do A, Esserman DA, Krishnan S et al. Excess weight gain after cure of hepatitis C infection with direct-acting antivirals. J Gen Intern Med 2020; 35(7): 2025–2034. doi: 10.1007/s11606-020-05782-6.
43. Pítová V, Fraňková S, Holinka M et al. PNPLA3 I148M variant is the main driver of weight gain after hepatitis C cure. Sci Rep 2025; 15(1): 25543. doi: 10.1038/s41598-025-11085-9.
44. Eslam M, Newsome PN, Sarin SK et al. A new definition for metabolic dysfunction-associated fatty liver disease: an international expert consensus statement. J Hepatol 2020; 73(1): 202–209. doi: 10.1016/j.jhep.2020.03.039.
45. Bugianesi E, Salamone F, Negro F. The interaction of metabolic factors with HCV infection: does it matter? J Hepatol 2012; 56(Suppl 1): S56–S65. doi: 10.1016/S0168-8278(12)60007-5.
46. Dawood AA, Nooh MZ, Elgamal AA. Factors associated with improved glycemic control by direct-acting antiviral agent treatment in Egyptian type 2 betes mellitus patients with chronic hepatitis C genotype 4. Diabetes Metab J 2017; 41(4): 316–321. doi: 10.4093/dmj.2017.41.4.316.