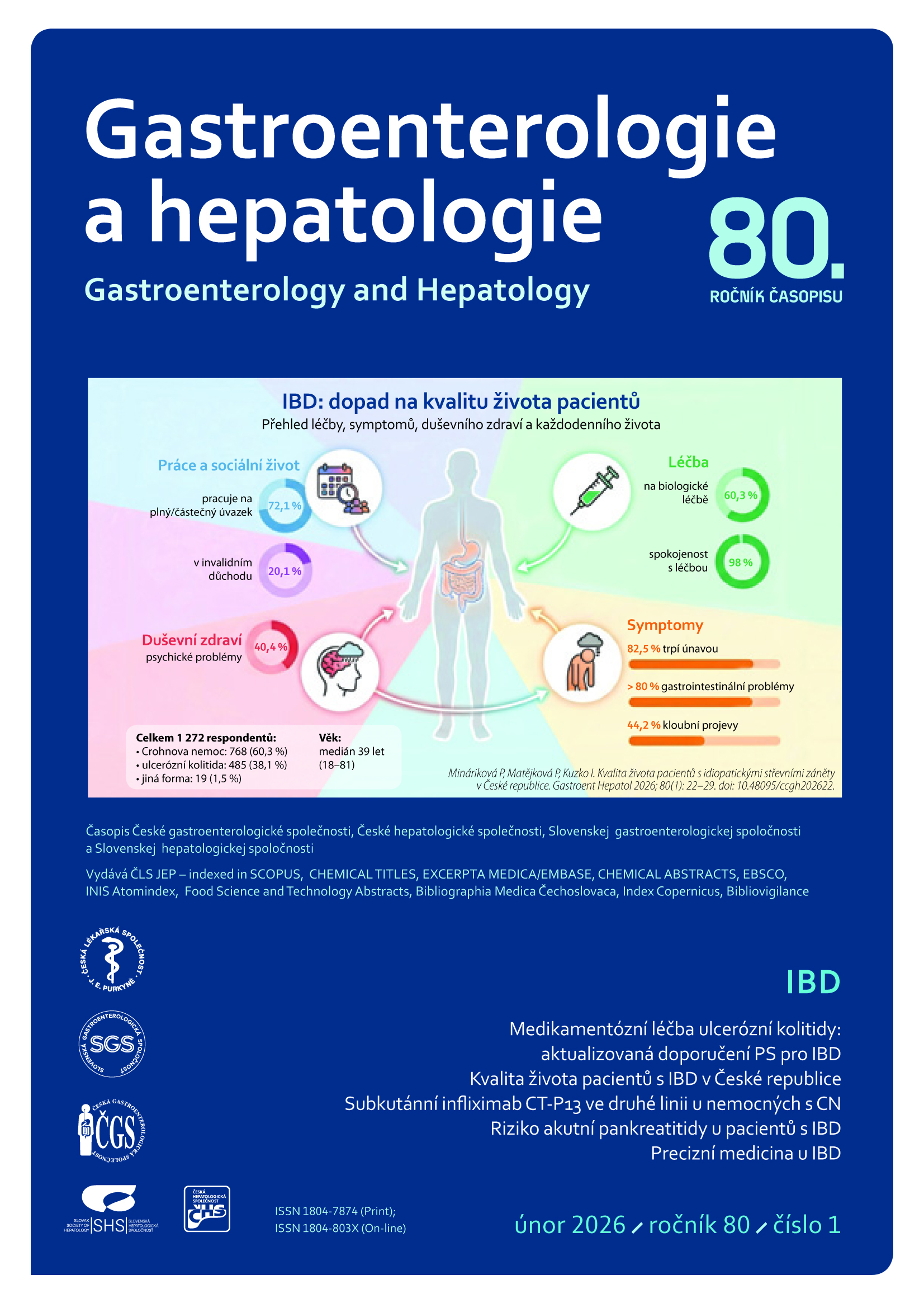
Abstract
Gastrointestinal bleeding (GIB) occurs more frequently and has greater clinical impact in patients with chronic kidney disease (CKD) than in patients without kidney disease. Current data prove that advanced stages of CKD, especially chronic renal failure (CRF), are independent risk factors for GIB. Hemodialysis treatment is a specific independent factor for GIB in CKD patients. Increased risk of GIB in CKD patients manifests by higher incidence and length of hospitalizations and higher mortality. The clinical condition of patients with advanced CKD and especially with CRF is usually more serious compared with patients without kidney disease. Moreover, it has been shown that the ability to provide these patients with an efficient treatment, especially early endoscopy, is decreased, which is partly associated with increased mortality. These findings suggest that greater attention should be paid to the topic of GIT in order to improve the outcomes and decrease the mortality of these patients.
This article is an English translation of the original Czech publication. The translation was generated with the assistance of artificial intelligence and has been reviewed and approved by the authors. The original version remains the authoritative source.
Introduction
Gastrointestinal bleeding (GIB) is generally a serious clinical problem, but it occurs more frequently in patients with chronic kidney disease (CKD) and can have more serious clinical consequences than in other patients. CKD is defined as a structural or functional abnormality of the kidneys lasting longer than 3 months and affecting the patient's health [1]. Epidemiological studies show that nearly 10% of the population has CKD, with a smaller proportion of patients having the most severe degree of functional impairment – chronic renal failure (CRF) – requiring some form of renal replacement therapy (dialysis or kidney transplantation) [1]. Various aspects of gastrointestinal problems and GIB in patients with CKD have not yet been systematically addressed, as evidenced, among other things, by the limited number of studies devoted to this topic [2].
Factors potentially influencing gastrointestinal bleeding in patients with CKD
Patients with CKD may experience certain circumstances that contribute to an increased incidence of GIB, its severity, and possibly more difficult treatment. A key factor is the fact that CKD is a complex metabolic disorder that affects not only kidney function but also the activity of other organs. Particularly significant is the high incidence of cardiovascular disease in more advanced stages of CKD [3].
Immediate effect of CKD on the GI mucosal barrier
In patients with CKD, an increased incidence of pathological GI mucosal lesions can be observed, which may predispose to GIB. In patients with CRF treated with regular dialysis, endoscopy has shown a high incidence of inflammatory changes and erosion of the mucosa of the esophagus, stomach, and duodenum, the development of which may be influenced to varying degrees by factors such as systemic and local circulatory disorders, elevated gastrin levels, retention of nitrogenous catabolites, and inflammatory changes [4]. According to some opinions, insufficient excretion of nitrogenous catabolites by the kidneys and their retention in the body may disrupt the intestinal barrier [5].
Specific features of CKD affecting the occurrence of GI lesions, their diagnosis and treatment
In the vast majority of patients with advanced chronic renal insufficiency (CRI) and CRF, pathophysiological processes typically associated with a decline in kidney function gradually worsen, resulting in the clinical manifestation of uremic syndrome. Anemia worsens, arterial hypertension becomes more pronounced, signs of hyperhydration appear, and patient care at this stage of the disease is usually quite demanding. On the one hand, the symptoms and manifestations of GI disease may be more frequent and severe at this stage of CKD, but diagnostic and therapeutic interventions may be associated with an increased risk compared to patients with normal kidney function or mild CRI.
GI disease and symptoms in patients with CKD – specific effect of underlying kidney disease
Although the pathophysiological processes and development of clinical manifestations of CKD are essentially uniform in all patients with CKD, the clinical picture of CKD and GI symptoms can be significantly influenced by specific extra-renal manifestations of certain so-called systemic diseases – typically, for example, many patients with diabetes mellitus (DM) develop digestive problems as a result of autonomic neuropathy. In some kidney diseases, the development of GI symptoms in patients with advanced CKD may also be closely linked to the type of treatment provided – for example, in many patients with immunopathological kidney disease, immunosuppressive therapy (often containing corticosteroids) is inherently associated with an increased risk of developing stomach ulcers and GIB.
GI diseases and symptoms in patients with CKD – specific effect of dialysis treatment and kidney transplantation
In patients with CKD in the end-stage renal disease (CRF) stage, the function of the original kidneys is replaced by regular dialysis treatment or kidney transplantation. The use of both methods is associated to a certain extent with some specific risks related to the particular type of treatment. In the Czech Republic, most European countries, and the US, hemodialysis (HD) is clearly the predominant type of dialysis treatment, and the alternative to HD treatment – peritoneal dialysis – is usually used in less than 10% of patients treated with dialysis. In terms of possible risk factors for GI symptoms, or rather the risk of GIB, a specific risk factor in HD treatment is primarily the need to ensure the patency of the vascular shunt for HD treatment (which is the most common vascular access in HD patients) with antiplatelet therapy and to prevent coagulation during HD treatment with anticoagulant therapy (currently most often in the form of a single dose of low molecular weight heparin before starting HD treatment). In kidney transplant patients, GI symptoms are common and are often related to the immunosuppressive treatment necessary in transplant patients or to infectious complications.
Sources of bleeding in patients with CKD
Precise data on the sources of bleeding are not known, partly because most studies evaluating the incidence of GIB in patients with CKD do not always distinguish between upper GI (UGIT) and lower GI (LGIT) bleeding. UGIT bleeding is generally reported as the clearly predominant type of bleeding, with LGIT bleeding being less common. Some data suggest that this distribution may be changing somewhat, at least in patients with CKD, with a decrease in UGIT bleeding due to the standard use of proton pump inhibitors for peptic ulcer disease (PUD) and Helicobacter pylori eradication, and conversely, an increased incidence of LGIT angiodysplasia and its complications in patients with CKD, especially in patients undergoing HD [6–8]. Various causes may contribute to the occurrence of angiodysplasia in LGIT in patients with CKD, including the older age of patients with CKD, antiplatelet and anticoagulant therapy, etc. [9].
Current evidence of gastrointestinal bleeding in patients with CKD – findings from clinical studies
There is no doubt that GIB in patients with CKD is a serious clinical event associated with high morbidity and mortality. However, there is currently a relatively limited amount of reliable data available on the incidence of GIB, factors contributing to GIB, and other important data related to this complication in the CKD patient population. Two large studies addressing this issue have recently been published. Lin et al. published a meta-analysis of 22 studies published by 2023 from publicly available databases (MedLine, Embase) related to the topic of GIB in patients with CKD; the total number of patients with CKD in this meta-analysis exceeded 7.8 million [10]. He et al. published a retrospective analysis of data from the US national database for the period 2016–2019, also related to the topic of GIB in patients with advanced CKD (CKD stages G4–5); the number of patients exceeded 2.1 million [11].
Incidence of gastrointestinal bleeding in patients with CKD
A recent large meta-analysis of studies on the incidence of GIB in patients with CKD shows that the incidence of GIB in the CKD patient population was 2.2% in published studies, and the incidence of GIB related to angiodysplasia was 0.4% [10]. Some studies have shown a significantly higher incidence of GIB in patients with CKD undergoing regular dialysis treatment compared to patients with CKD not undergoing dialysis treatment – specifically, in the study by Little et al. 5.74 per 100 person-years vs. 2.42 per 100 person-years [12]. In another study, the risk of GIB in patients with CKD was also significantly increased (by 35%) compared to GIB in patients without CKD [13].
In a population of patients with CKD who underwent endoscopic examination, GIB was found in 35.8% of patients, GIB associated with peptic ulcer in 14.2% of patients, and bleeding associated with angiodysplasia in 14.3% of patients [10].
Hospitalization and mortality associated with gastrointestinal bleeding in patients with CKD
A meta-analysis of recent studies showed that patients with CKD in the CRF stage (3.4%) and patients with CKD in the advanced CHRI stage (3.2%) were significantly more likely to be hospitalized for GIB than patients without CKD (2.2%) [10]. This increased incidence of hospitalizations in patients with CRF was confirmed in a recent retrospective analysis from the USA [11]. In terms of mortality among patients hospitalized for GIB, the highest mortality was observed in patients with CRF (3.1%), followed by patients with advanced CHRI (3%) and finally patients after kidney transplantation (2%) – i.e., values significantly higher than the mortality of 1.7% among patients without CKD [10].
Sources of bleeding
In a study of GIB in patients with CKD undergoing hemodialysis treatment, the ratio of UGIT to LGIT bleeding episodes was 65.2% to 33.7% [14]. In a recent retrospective analysis from the USA, the most common cause of UGIT bleeding in the total number of patients was (excluding bleeding from esophageal varices; 38.7%), followed by unspecified GIB (melena, hematemesis, or unspecified bleeding without specification of the source; 37%), LGIT bleeding (22.9%), and esophageal UGIT bleeding (1.4%). When the category of unspecified GIB was excluded, the most common types of bleeding in the group of patients without kidney disease and patients after kidney transplantation were gastroduodenal bleeding, diverticular bleeding, and bleeding from esophageal varices, while in patients with advanced CHRI and CRF, the most common types of bleeding were gastroduodenal bleeding, bleeding from angiodysplasia, and diverticular bleeding [11].
Factors influencing gastrointestinal bleeding in patients with CKD
Several specific factors influence the (higher) incidence of GIB in patients with CKD compared to patients without CKD:
- The most significant specific factor influencing bleeding in patients with CKD is undoubtedly regular dialysis treatment (odds ratio: OR 14.48; at a 95% confidence interval: CI 4.96–42.32) [10]. The causes of this high incidence have not been fully clarified, but it can be assumed that the overall serious clinical condition of patients and the higher probability of bleeding due to the fact that most patients treated with dialysis or hemodialysis receive permanent or temporary antiplatelet and/or anticoagulant therapy play a major role [15].
- DM is also a significant predictor of GIB (OR 1.03; 95% CI 1.05–1.05) [9]. DM is a systemic disease with an extremely adverse impact on the function of various organs, including the kidneys, GI, but also the cardiovascular system, etc. The increased risk of GIB is apparently multifactorial.
- Other proven predictors of GIB in patients with CKD were older age (OR 1.03; 95% CI 1.02–1.05), PUD (OR 1.53; 95% CI 1.03–2.26), and liver cirrhosis (OR 1.73; 95% CI 1.45–3.10) [10].
- The resulting picture of an increased incidence of GIB in patients with CKD probably reflects not only the actual increased risk of GIB in patients with CKD as such, but also the lower ability to manage these complications [16]. A retrospective study from the USA clearly showed that the overall number of endoscopic procedures in patients with CRF was significantly lower compared to other patient groups (OR 0.97; 95% CI 0.93–0.99; p = 0.04), that early endoscopic procedures and other interventional procedures (arterial embolization and surgical interventions) are less common in patients with severe kidney disease, and that the reduced ability to provide these patients with early intervention is associated with a higher risk of mortality [11]. Early endoscopy (within 24 hours of admission to hospital) was less common in patients with CKD compared to patients without kidney disease (for early esophagogastroduodenoscopy (EGD; OR 0.97; 95% CI 0.80–0.85; p < 0.001); for early colonoscopy (CSC; OR 0.67; 95% CI, 0.63–0.71; p < 0.001), while delayed endoscopy (after 48 hours of admission) was significantly more common (for delayed EGD OR 1.2; 95% CI 1.16–1.23; p < 0.001; for early CSC OR 1.44; 95% CI 1.39–1.5; p < 0.001) [11].
A similar trend was observed in patients with advanced CHRI; conversely, in patients after kidney transplantation, the proportion of early and delayed procedures was similar to that in the general population [11]. A lower proportion of early endoscopic procedures and a higher number of delayed procedures were associated with higher mortality in patients with CRF, and similar conclusions were evident from other studies [11,16]. The overall clinical condition of patients with CRF was the most severe (compared to patients with CHRI, kidney transplant patients, and the general population), which was reflected in a longer length of hospital stay, a higher number of cases of mechanical ventilation, vasopressor support, and blood transfusions [11]. Anticoagulant and antiplatelet therapy were shown in this study to be independent factors for the incidence of hospitalizations of patients with GIB [11].
Conclusions
Based on the available recent evidence, it can be concluded that advanced stages of CKD and especially CRF are significant independent factors for GIB. Regular hemodialysis treatment is a specific independent factor for GIB. GIB in these patients results in increased frequency and length of hospitalizations and increased mortality (Tab. 1).
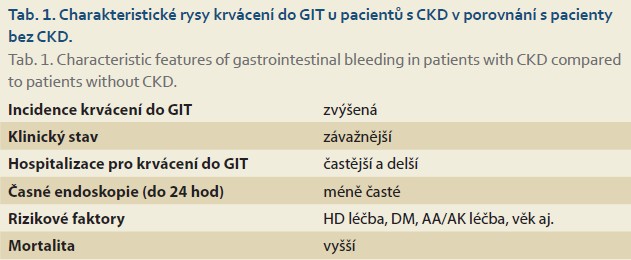
The overall clinical condition of patients with CRF admitted to hospital due to GIB is usually more serious than in other patients, which can be documented by longer hospital stays, a higher number of mechanical ventilations, a higher number of blood transfusions, etc.
In addition to the fact that patients in advanced stages of CKD (CRF and CHRI) are in a more serious clinical condition than the general population, it is clear that the ability to provide these patients with effective treatment, especially early endoscopic treatment, is reduced. This is likely to be partly reflected in the increased mortality rate among these patients.
Findings on GIB in patients with CKD suggest that this issue needs to be given increased attention in order to improve treatment outcomes and reduce mortality in these patients. One way to achieve this is to improve the awareness of the professional community and to analyze in more detail the individual causes contributing to this unsatisfactory situation. It is also necessary to monitor any changes in the causes of bleeding in patients with CKD and respond to these developments. In specific practice, for example, in patients with CKD, it is necessary to carefully consider the necessary degree of antiplatelet and anticoagulant therapy in order to minimize the risk of bleeding, look for possible bleeding, limit the use of substances that increase the risk of bleeding, provide drugs that are effective in treating or preventing ulcerative PUD, and in the case of unexplained worsening of anemia, ensure timely diagnosis and treatment [11,17–19].
Seznam zkratek
DM diabetes mellitus
CHR chronická renální insuficience
HGIT horní gastrointestinální trakt
DGIT dolní gastrointestinální trakt
GDCH gastroduodenální vředová choroba
EGD ezofagogastroduodenoskopie
GIT gastrointestinální trakt
CKD chronické onemocnění ledvin
HD hemodialýza
AA antiagregační léčba
AK antikoagulační léčba
Doručeno/Submitted: 23. 1. 2026
Přijato/Accepted: 30. 1. 2026
Korespondenční autor
prof. MUDr. Miroslav Merta, CSc.
Nefrologická ambulance
Affidea Nefromed s.r.o
Africká 687/36
160 00 Praha 6
mirek.merta@seznam.cz
Literatura
1. Bikbov B, Purcell CA, Levey AS et al. Global, regional, and national burden of chronic kidney disease, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 2020; 395(10225): 709–733. doi: 10.1016/S0140-6736(20)30045-3.
2. Teplan V, Marečková O, Lukáš M. Onemocnění gastrointestinálního traktu a choroby ledvin. Gastroent Hepatol 2018; 72(1): 50–57. doi: 10.14735/amgh201850.
3. Zoccali C, Vanholder R, Massy ZA et al. The systemic nature of CKD. Nat Rev Nephrol 2017; 13(6): 344–358. doi: 10.1038/nrneph.2017.52.
4. Krishnan A, Venkataraman J. Gastrointestinal evaluation in chronic kidney diseases. J Nephrol Ther 2011; 1(3): 110. doi: 10.4172/2161-0959.1000110.
5. Chung SY, Barnes JL, Astroth KS. Gastrointestinal microbiota in patients with chronic kidney disease: a systematic review. Adv Nutr 2019; 10(5): 888–901. doi: 10.1093/advances/nmz028.
6. Wuerth BA, Rockey DC. Changing epidemiology of upper gastrointestinal hemorrhage in the last decade: a nationwide analysis. Dig Dis Sci 2018; 63(5): 1286–1293. doi: 10.1007/s10620-017-4882-6.
7. He M, Shah AD, Wang Y et al. Impact of CKD/kidney failure on angiodysplasia-related gastroinestinal bleeding: incidence, outcomes and readmission risk in a national cohort. Kidney360 2025; 6(10): 1689–1701. doi: 10.34067/KID.0000000901.
8. Barkun AN, Almadi M, Kuipers EJ et al. Management of nonvariceal upper gastrointestinal bleeding: guideline recommendations from the International Consensus Group. Ann Intern Med 2019; 171(11): 805–822. doi: 10.7326/M19-1795.
9. Sami SS, Al-Araji SA, Ragunath K. Review article: gastrointestinal angiodysplasia – pathogenesis, diagnosis and management. Aliment Pharmacol Ther 2014; 39(1): 15–34. doi: 10.1111/apt.12527.
10. Lin Y, Li C, Waters D et al. Gastrointestinal bleeding in chronic kidney disease patients: a systematic review and meta-analysis. Ren Fail 2023; 45(2): 2276908. doi: 10.1080/0886022X.2023.2276908.
11. He M, Desai S, Wang Y et al. Inpatient outcomes of gastrointestinal bleeding in advanced CKD and kidney transplant recipients: a national analysis 2016–2019. Kidney360 2025; 6(3): 379–390. doi: 10.34067/KID.0000000662.
12. Little DJ, Arnold M, Hedman K et al. Rates of adverse clinical events in patients with chronic kidney disease: analysis of electronic health records from the UK clinical practice research datalink linked to hospital data. BMC Nephrol 2023; 24(1): 91. doi: 10.1186/s12882-023-03119-z.
13. Mahady SE, Polekhina G, Woods RL et al. Association between CKD and major hemorrhage in older person: data from the aspirin in reducing events in the elderly randomized trial. Kidney Int Rep 2023; 8(4): 737–745. doi: 10.1016/j.ekir.2023.01.012.
14. Trivedi H, Yang J, Szabo A. Gastrointestinal bleeding in patients on long-term dialysis. J Nephrol 2015; 28(2): 235–243. doi: 10.1007/s40620-014-0132-6.
15. Huang CH, Chao JY, Ling TC et al. Effect of dialysis modalities on risk of hospitalization for gastrointestinal bleeding. Sci Rep 2023; 13(1): 52. doi: 10.1038/s41598-022-26476-5.
16. Garg R, Parikh MP, Chadalvada P et al. Lower rates of endoscopy and higher mortality in end-stage renal disease patients with gastrointestinal bleeding: a propensity matched National Study. J Gastroenterol Hepatol 2022; 37(3): 584–591. doi: 10.1111/jgh.15771.
17.Tai FWD, McAlindon ME. Non-steroidal anti-inflammatory drugs and the gastrointestinal tract. Clin Med 2021; 21(2): 131–134. doi: 10.7861/clinmed.2021-0039.
18. Jones A, Swan D, Lisman T et al. Anticoagulation in chronic kidney disease and future perspectives. J Thromb Haemost 2024; 22(2): 323–336. doi: 10.1016/j.jtha.2023.09.020.
19. Elrggal ME, Naga YS. Gastrointestinal bleeding in patients with CKD: the renalism continues! Kidney 2025; 6(3): 337–339. doi: 10.34067/KID.0000000686.