Abstrakt
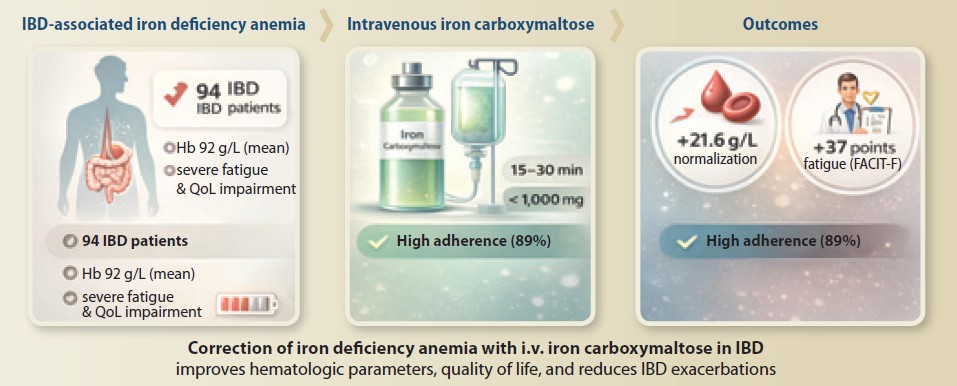
Úvod: Anémia z nedostatku železa (IDA) je častá mimokrevná manifestácia zápalových črevných ochorení (IBD), ktorá výrazne zhoršuje kvalitu života. Táto štú hodnotila účinnosť intravenóznej železovej karboxymaltózy (ICM) pri korekcii IDA a jej vplyv na kvalitu života, adherenciu k liečbe a frekvenciu exacerbácií ochorenia u pacientov s IBD. Metódy: Prospektívna jednocentrová štú zahŕňala 94 dospelých pacientov s IBD a potvrdenou IDA. Účastníci dostali intravenóznu ICM (dávka vypočítaná podľa Ganzoniho vzorca) a boli sledovaní po dobu 6 mesiacov. Primárne výsledky zahŕňali normalizáciu hemoglobínu (Hb), hodnoty kvality života (dotazník IBDQ), adherenciu k liečbe a miery exacerbácií ochorenia. Výsledky: Podanie ICM viedlo k významnému zvýšeniu hladín Hb (z 92,1 ± 10,8 g/l na 113,7 ± 11,0 g/l; p < 0,0001) a sérového železa (z 4,6 ± 2,1 µmol/l na 20,3 ± 9,1 µmol/L; p < 0,0001). Skóre kvality života sa výrazne zlepšili (IBDQ z 125 ± 24 na 162 ± 19; p < 0,001). Adherencia k liečbe bola 89,4 % a frekvencia exacerbácií IBD klesla o 52,3 % v porovnaní s pred liečbou. Záver: Úspešná korekcia anémie s použitím ICM u pacientov s IBD výrazne zlepšuje kvalitu života, zvyšuje adherenciu k liečbe a znižuje frekvenciu exacerbácií ochorenia, čo podporuje dôležitosť agresívneho manažmentu anémie v tejto populácii.
Introduction
Iron deficiency anemia (IDA) represents one of the most prevalent extraintestinal manifestations of inflammatory bowel disease (IBD), affecting approximately 60–80% of patients during the disease course. The pathogenesis of IDA in IBD is multifactorial, resulting from chronic intestinal blood loss, impaired iron absorption due to mucosal inflammation, and nutritional deficiencies stemming from dietary restrictions. The clinical significance of IDA extends beyond hematological parameters, as it substantially contributes to profound fatigue, reduced exercise capacity, and impaired quality of life frequently reported by IBD patients.
The complex interplay between iron metabolism and chronic inflammation creates particular challenges for anemia management in IBD. Inflammation elevates hepcidin levels, which inhibits both intestinal iron absorption and iron release from macrophage stores. This phenomenon explains the limited efficacy of oral iron supplementation in active IBD, as demonstrated by high rates of intolerance and treatment failure [1]. Additionally, oral iron may exacerbate gastrointestinal symptoms and potentially worsen intestinal inflammation in some patients, further complicating management.
Intravenous iron preparations bypass the hepcidin-meted blockade, offering a more effective approach for iron repletion in inflammatory conditions. Among available options, iron carboxymaltose (ICM) has emerged as a particularly valuable formulation due to its ability to administer high doses (up to 1,000 mg) in brief infusion sessions (15–30 minutes), enhancing patient convenience and treatment adherence [2]. Previous studies in other clinical contexts, including colorectal cancer and pregnancy, have demonstrated the efficacy of ICM in rapidly correcting hemoglobin and iron parameters [3,4].
Despite established hematological efficacy, important knowledge gaps remain regarding the broader impact of successful anemia correction with ICM in IBD patients. Limited evidence exists on how ICM-meted anemia correction influences quality of life, treatment adherence, and most importantly, the natural history of IBD, including exacerbation frequency. Previous research has highlighted that treatment side effects significantly compromise adherence to anemia therapy, but whether the favorable safety profile of ICM translates to better adherence in IBD populations requires investigation.
This study tests the hypothesis that successful IDA correction with intravenous ICM in IBD patients significantly improves quality of life measures, enhances treatment adherence, and reduces the frequency of disease exacerbations. We propose that comprehensive anemia management represents not only a supportive care measure, but potentially acts as a modifiable factor influencing IBD disease activity [5–7].
Materials and methods
Study design and patient population
A prospective, single-center, observational study was conducted between June 2023 and December 2024 at a tertiary IBD referral center. The study protocol was approved by the institutional ethics committee, and all participants provided written informed consent.
The study enrolled 94 adult patients (age ≥ 18 years) with established gnosis of IBD (Crohn‘s disease or ulcerative colitis) according to standard clinical, endoscopic, and histopathological criteria. Participants had confirmed iron deficiency anemia, defined by hemoglobin (Hb) levels < 120 g/L for women and < 130 g/L for men, in combination with ferritin levels < 30 μg/L or transferrin saturation (TSAT) < 16%. For patients with active inflammation (C-reactive protein > 5 mg/L), a ferritin cutoff < 100 μg/L was applied to account for inflammation-driven ferritin elevation.
Exclusion criteria included: anemia from causes other than iron deficiency (B12/folate deficiency, hemolytic anemia), severe comorbidities (heart failure NYHA III–IV, chronic kidney disease stage 4–5), active malignancy, pregnancy or lactation, history of hypersensitivity to intravenous iron preparations, and erythrocyte transfusion within 4 weeks prior to enrollment.
Intervention protocol
All participants received intravenous iron carboxymaltose (Ferinject®) administered according to the Ganzoni formula: total iron deficit (mg) = body weight (kg) × (target Hb – actual Hb) (g/L) × 0.24 + depot iron 500 mg. The maximum single dose did not exceed 1,000 mg, and was administered as a slow intravenous injection or brief infusion (15–30 minutes) according to manufacturer recommendations. Doses exceeding 1,000 mg were divided with at least 7-day intervals between administrations.
Concomitant IBD medications (5-aminosalicylates, immunomodulators, biologics) remained stable throughout the study period unless modification was clinically necessitated by disease exacerbation.
Assessment methods
Hematological parameters: Hemoglobin (Hb), serum ferritin, transferrin saturation (TSAT), serum iron, and complete blood count were assessed at baseline, 2 weeks, 4 weeks, 12 weeks, and 24 weeks post-treatment.
Quality of life measurement: Disease-specific quality of life was evaluated using the Inflammatory Bowel Disease Questionnaire (IBDQ), a validated 32-item instrument covering bowel symptoms, systemic symptoms, emotional function, and social function. The IBDQ was administered at baseline and 12 weeks. Additionally, fatigue was assessed using the Functional Assessment of Chronic Illness Therapy-Fatigue (FACIT-F) scale at the same timepoints.
Treatment adherence: Adherence to ICM therapy was calculated as the percentage of patients who completed all prescribed iron doses. For adherence to overall IBD treatment, the Medication Adherence Report Scale (MARS-5) was administered at baseline and 12 weeks.
Disease exacerbation monitoring: IBD exacerbations were defined as requiring corticosteroid therapy, hospitalization, or surgical intervention related to IBD, or escalation of maintenance medication. Exacerbation frequency during the 6-month follow-up period was compared to the 6-month pre-treatment period for each patient.
Safety assessment: Adverse events were recorded throughout the study period and categorized following the Common Terminology Criteria for Adverse Events (CTCAE) version 5.
Statistical analysis
Statistical analysis was performed using SPSS version 24.0. Continuous variables were expressed as mean ± standard deviation or men with interquartile range based on distribution normality, assessed by the Shapiro-Wilk test. Categorical variables were presented as frequencies and percentages. Within-group comparisons employed the paired t-test or Wilcoxon signed- -rank test for continuous variables, and McNemar‘s test for categorical variables. Between-group comparisons used the independent t-test or Mann-Whitney U test for continuous variables, and Chi-square or Fisher‘s exact test for categorical variables. Correlation analyses utilized Pearson or Spearman coefficients. A two-sided P-value < 0.05 was considered statistically significant.
Results
Baseline demographic and clinical characteristics
A total of 94 patients completed the study protocol from initial enrollment to final follow-up. The baseline characteristics of the study population are summarized in Tab. 1.
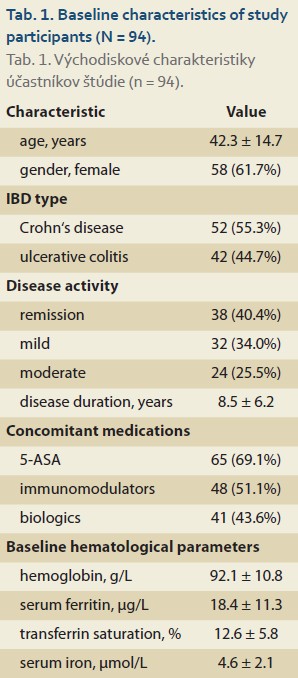
The study population comprised of a representative IBD cohort with slightly more Crohn‘s disease patients (55.3%) and a female predominance (61.7%), reflecting a higher prevalence of IDA in women of reproductive age. Most patients had mild (34.0%) or moderate (25.5%) disease activity at enrollment, with a mean disease duration of 8.5 years, indicating established IBD.
Hematological efficacy
Intravenous ICM administration resulted in significant improvements in all hematological parameters. As illustrated in Tab. 2, hemoglobin levels increased progressively from a baseline mean of 92.1 ± 10.8 g/L to 113.7 ± 11.0 g/L at week 4 (P < 0.0001), with sustained improvement through week 24. The mean hemoglobin increase was 21.6 ± 11.5 g/L (P < 0.0001), consistent with previous studies of ICM in other patient populations.
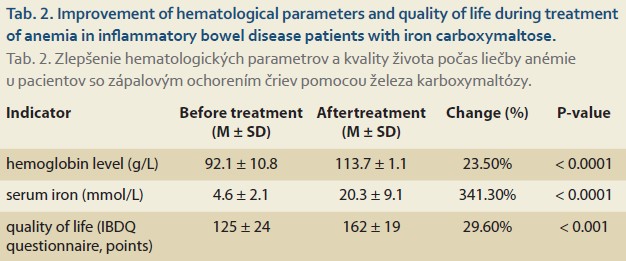
Iron storage parameters demonstrated even more pronounced improvement. Serum ferritin levels increased from 18.4 ± 11.3 μg/L to 128.7 ± 65.4 μg/L at week 4 (P < 0.0001), indicating successful repletion of iron stores. Transferrin saturation improved from 12.6 ± 5.8% to 28.4 ± 11.2% (P < 0.0001), and serum iron levels increased from 4.6 ± 2.1 μmol/L to 20.3 ± 9.1 μmol/L (P < 0.0001).
The response rate was high, with 87 patients (92.6%) achieving hemoglobin normalization (Hb >120 g/L for women, >130 g/L for men) by week 12. The remaining seven patients had significant comorbidities contributing to persistent anemia (chronic kidney disease, N = 3; active inflammation, N = 4).
Quality of life outcomes
Successful anemia correction translated to significant improvements in quality--of-life measures. The total IBDQ score increased from 125 ± 24 at baseline to 162 ± 19 at week 12 (P < 0.001), exceeding the minimal clinically important difference of 16 points for this instrument. All IBDQ domains demonstrated significant improvement (Tab. 3).
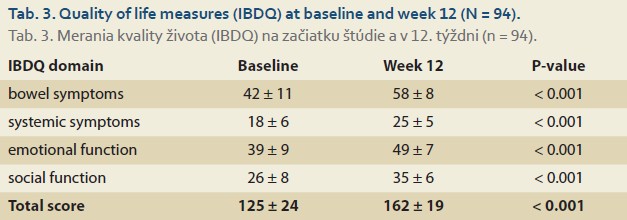
Fatigue scores improved substantially, with the FACIT-F scale increasing from 24.5 ± 8.3 to 35.2 ± 7.1 (P < 0.001). Subgroup analysis revealed that patients with more severe baseline anemia (Hb < 100 g/L) experienced the greatest magnitude of improvement in quality-of-life measures (mean IBDQ increase of 43.2 points versus 28.7 points for mild anemia, P = 0.012).
Treatment adherence
Treatment adherence to ICM was exceptionally high, with 84 of 94 patients (89.4%) completing all prescribed iron doses. This exceeds typical adherence rates for oral iron supplementation, which previous studies report between 50 and 70% in chronic conditions. Furthermore, adherence to overall IBD medication, as measured by the MARS-5 questionnaire, improved significantly from 76.3% at baseline to 85.7% at week 12 (P = 0.008).
Patients cited the convenience of single or dual infusion sessions (vs. daily oral supplementation) and the absence of significant gastrointestinal side effects as primary reasons for high adherence to ICM. Notably, only 6 patients (6.4%) reported treatment-related adverse events, all of which were mild (CTCAE grade 1–2) and transient, including two cases of self-limiting headache, three cases of mild nausea, and one case of transient hypotension during infusion.
Disease exacerbation frequency
A particularly noteworthy finding was the significant reduction in IBD exacerbation frequency following successful anemia correction. During the 6-month follow-up period after ICM treatment, 18 patients (19.1%) experienced disease exacerbations, compared to 37 patients (39.4%) during the 6-month pre-treatment period (P = 0.003). This represents a 52.3% relative reduction in exacerbation frequency.
The time to first exacerbation was significantly prolonged in the post-treatment period (men not reached versus 18.7 weeks in pre-treatment period, P = 0.004 by log-rank test). Subgroup analysis suggested that patients with more pronounced hemoglobin improvement (> 20 g/L increase) experienced greater reduction in exacerbation risk (HR 0.42, 95% CI 0.23–0.78; P = 0.006).
Safety profile
The ICM intervention demonstrated a favorable safety profile, with only 6.4% of patients experiencing any adverse event, and no severe (grade 3–4) adverse events were recorded. This aligns with previous reports of ICM safety in other patient populations. No anaphylactic reactions occurred, and no patients discontinued treatment due to adverse events.
Discussion
This prospective study demonstrates that successful correction of iron deficiency anemia with intravenous iron carboxymaltose in IBD patients yields benefits extending far beyond hematological normalization. We observed significant improvements in the quality of life, enhanced treatment adherence, and notably, a substantial reduction in disease exacerbation frequency.
The hematological efficacy of ICM in our IBD cohort aligns with previous reports in other patient populations. The mean hemoglobin increase of 21.6 g/L and improvement in iron storage parameters are consistent with studies of ICM in colorectal cancer and pregnancy. The rapid response observed – with significant improvement within 2 weeks and normalization in most patients by 4 weeks – is clinically meaningful, particularly for patients experiencing profound fatigue and functional impairment.
The quality-of-life improvement in our study (mean IBDQ increase of 37 points) exceeds the minimal clinically important difference, representing one of the most substantial benefits associated with any intervention in IBD supportive care. This finding underscores that anemia contributes significantly to the symptom burden in IBD, independent of intestinal inflammation itself. The mechanism likely involves improved oxygen delivery to tissues, enhanced cellular metabolism, and reduced carc strain from chronic anemia. The particularly pronounced improvement in fatigue scores aligns with previous reports that iron deficiency without anemia can cause significant fatigue, suggesting that iron repletion benefits extend beyond correction of hemoglobin levels.
The high treatment adherence (89.4%) with ICM contrasts sharply with the suboptimal adherence typically observed with oral iron preparations. Previous research indicates that 45% of anemia patients experience treatment-related side effects, with higher prevalence in oral iron formulations. Gastrointestinal intolerance to oral iron is particularly problematic in IBD patients, who may already have sensitive digestive systems. The convenience of brief, infrequent ICM administrations likely contributed to the excellent adherence observed.
The most intriguing finding of our study is the 52.3% reduction in IBD exacerbation frequency following successful anemia correction. While observational in nature, this association suggests several potential mechanisms. Chronic anemia and tissue iron deficiency may directly exacerbate inflammation through increased oxidative stress or impaired immune function. Alternatively, improved overall well-being might enhance adherence to IBD maintenance therapies. The correlation between greater hemoglobin improvement and greater exacerbation reduction supports a potential dose-response relationship. This finding warrants confirmation in randomized controlled trials, but suggests that aggressive anemia management may modify the natural history of IBD.
Our study has several limitations. The single-center design may limit generalizability, even though our patient population represents a typical IBD cohort. The absence of a control group receiving placebo or oral iron prevents definitive causal conclusions regarding exacerbation reduction. However, using patients as their own historical control strengthens the observed association. Finally, the 6-month follow-up period precludes assessment of long-term outcomes.
The favorable safety profile of ICM in our study aligns with previous reports and supports its use as a first-line intravenous iron formulation in IBD patients requiring rapid iron repletion.
Conclusion
This study demonstrates that successful iron deficiency anemia correction with intravenous iron carboxymaltose in inflammatory bowel disease patients produces multifaceted benefits. Beyond the expected hematological normalization, we observed a significantly improved quality of life, enhanced treatment adherence, and substantially reduced disease exacerbation frequency.
These findings support these key clinical implications:
- Comprehensive anemia assessment should be integral to IBD management, with regular screening for iron deficiency even in non-anemic patients.
- Intravenous iron carboxymaltose represents an effective and well-tolerated option for iron repletion in IBD, particularly suited for patients with active inflammation, oral iron intolerance, or need for rapid correction.
- Successful anemia management may potentially modify the IBD disease course beyond symptomatic improvement, possibly reducing exacerbation frequency.
Future randomized controlled trials with longer follow-up are warranted to confirm the potential disease-modifying effect of anemia correction and to elucidate the underlying mechanisms. Additionally, comparative effectiveness research between different intravenous iron formulations in IBD populations would further refine treatment algorithms.
Acknowledgments
The authors thank the patients who participated in this study and the nursing staff for their dedicated care.
ORCID of authors
S. Dibraeva 0009-0007-5619-4560,
N. Iminova 0009-0006-9037-3696,
I. Atasheva 0009-0007-0391-9716,
R. Abdulgamidova 0009-0002-1912-0275,
R. Aigunova 0009-0001-8610-3988,
K. Magomedova 0009-0007-9185-7665,
M. Abakarova 0009-0004-0441-2587.
Submitted/Doručené: 23. 10. 2025
Accepted/Prijaté: 19. 11. 2025
Corresponding author
Salikhat Dibraeva, MD
Dagestan State Medical University
Lenin Square 1
367000 Makhachkala
Republic of Dagestan, Russia
dibraeva001@gmail.com
References
1. Moum B, Lindgren S. Iron deficiency and iron deficiency anemia in chronic disease – common, important, and treatable. J Clin Med 2025; 14(13): 4519. doi: 10.3390/jcm14134519.
2. Ciont C, Pop RM, Pop L et al. Impact of side effects on anemia therapy compliance. Nutrients 2025; 17(9): 1485. doi: 10.3390/nu17091485.
3. Kornev DO, Moskalenko AN, Kuzmina ES et al. Ferric carboxymaltose in the treatment of anemia in patients with locally advanced colon cancer. Creat Surg Oncol 2025; 15(1): 12–18. doi: 10.24060/2076-3093-2025-15-1-12-18.
4. Bartsits MK, Safronenko AV, Gantsgorn EV et al. Evaluation of the effectiveness of the use of iron carboxymaltosate in late pregnancy and in the postpartum period. South Russian J Ther Pract 2023; 4(4): 83–88. doi: 10.21886/2712-8156-2023-4-4-83-88.
5. Iron deficiency conditions in a doctor’s practice: news and an interdisciplinary approach. Effect Pharmacother 2021; 17(43): 42–50.
6. Clinical Guidelines: iron deficiency anemia. Russian Ministry of Health. 2024 [online]. Available from: https: //diseases.medelement.com/disease/железодефицитная-анемия-кр-рф-2024/18265.
7. Iron deficiency in chronic kidney disease: multiple consequences and correction algorithm. Effect Pharmacother 2024; 20(21).